Histone demethylase LSD1 deficiency during high-salt diet is associated with enhanced vascular contraction, altered NO-cGMP relaxation pathway, and hypertension
- PMID: 21873498
- PMCID: PMC3213961
- DOI: 10.1152/ajpheart.00513.2011
Histone demethylase LSD1 deficiency during high-salt diet is associated with enhanced vascular contraction, altered NO-cGMP relaxation pathway, and hypertension
Abstract
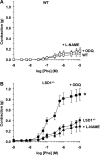
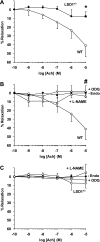
Histone methylation, a determinant of chromatin structure and gene transcription, was thought to be irreversible, but recent evidence suggests that lysine-specific demethylase-1 (LSD1, Kdm1a) induces demethylation of histone H3 lysine 4 (H3K4) or H3K9 and thereby alters gene transcription. We previously demonstrated a human LSD1 phenotype associated with salt-sensitive hypertension. To test the hypothesis that LSD1 plays a role in the regulation of blood pressure (BP) via vascular mechanisms and gene transcription, we measured BP and examined vascular function and endothelial nitric oxide (NO) synthase (eNOS) expression in thoracic aorta of male wild-type (WT) and heterozygous LSD1 knockout mice (LSD1(+/-)) fed either a liberal salt (HS; 4% NaCl) or restricted salt diet (LS; 0.08% NaCl). BP was higher in LSD1(+/-) than WT mice on the HS diet but not different between LSD1(+/-) and WT mice on the LS diet. Further examination of the mechanisms of this salt-sensitive hypertension in LSD1(+/-) mice on the HS diet demonstrated that plasma renin activity and plasma levels and urinary excretion of aldosterone were less in LSD1(+/-) than WT, suggesting suppressed renin-angiotensin-aldosterone system. In contrast, phenylephrine (Phe)-induced aortic contraction was greater in LSD1(+/-) than WT mice on the HS diet. Treatment of aortic rings with 1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one (ODQ; a blocker of guanylate cyclase) enhanced Phe contraction in LSD1(+/-) compared with WT mice on the HS diet. Acetylcholine (Ach)-induced relaxation was less in LSD1(+/-) than WT mice on the HS diet. Endothelium removal or pretreatment with N(ω)-nitro-L-arginine methyl ester (blocker of NOS) or ODQ abolished Ach-induced relaxation in aorta of WT but had minimal effect in LSD1(+/-). Vascular relaxation to sodium nitroprusside, an exogenous NO donor and guanylate cyclase activator, was decreased in LSD1(+/-) vs. WT mice on the HS diet. RT-PCR and Western blots revealed decreased eNOS mRNA expression and eNOS and guanylate cyclase protein in the heart and aorta of LSD1(+/-) compared with WT mice on HS diet. Thus, during the HS diet, LSD1 deficiency is associated with hypertension, enhanced vascular contraction, and reduced relaxation via NO-cGMP pathway. The data support a role for LSD1-mediated histone demethylation in the regulation of NOS/guanylate cyclase gene expression, vascular function, and BP during the HS diet.
Figures
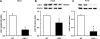

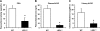
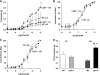
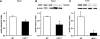

References
-
- Adler GK, Williams GH. Aldosterone: villain or protector? Hypertension 50: 31–32, 2007 - PubMed
-
- Cain AE, Khalil RA. Pathophysiology of essential hypertension: role of the pump, the vessel, and the kidney. Semin Nephrol 22: 3–16, 2002 - PubMed
-
- de Wardener HE, He FJ, MacGregor GA. Plasma sodium and hypertension. Kidney Int 66: 2454–2466, 2004 - PubMed
-
- Fleming I, Busse R. NO: the primary EDRF. J Mol Cell Cardiol 31: 5–14, 1999 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- K23 HL084236/HL/NHLBI NIH HHS/United States
- HL-65998/HL/NHLBI NIH HHS/United States
- R01 HL065998/HL/NHLBI NIH HHS/United States
- HD-60702/HD/NICHD NIH HHS/United States
- R01 GM078458/GM/NIGMS NIH HHS/United States
- R21 HL098724/HL/NHLBI NIH HHS/United States
- R01 HL069208/HL/NHLBI NIH HHS/United States
- GM-078458/GM/NIGMS NIH HHS/United States
- K23-HL-084236/HL/NHLBI NIH HHS/United States
- HL-104032/HL/NHLBI NIH HHS/United States
- HL-69208/HL/NHLBI NIH HHS/United States
- R01 HL104032/HL/NHLBI NIH HHS/United States
- HL-98724/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

