Innate immune recognition of bacterial ligands by NAIPs determines inflammasome specificity
- PMID: 21874021
- PMCID: PMC3184209
- DOI: 10.1038/nature10394
Innate immune recognition of bacterial ligands by NAIPs determines inflammasome specificity
Abstract
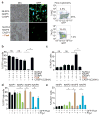
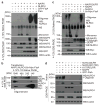
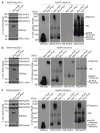
Inflammasomes are a family of cytosolic multiprotein complexes that initiate innate immune responses to pathogenic microbes by activating the caspase 1 protease. Although genetic data support a critical role for inflammasomes in immune defence and inflammatory diseases, the molecular basis by which individual inflammasomes respond to specific stimuli remains poorly understood. The inflammasome that contains the NLRC4 (NLR family, CARD domain containing 4) protein was previously shown to be activated in response to two distinct bacterial proteins, flagellin and PrgJ, a conserved component of pathogen-associated type III secretion systems. However, direct binding between NLRC4 and flagellin or PrgJ has never been demonstrated. A homologue of NLRC4, NAIP5 (NLR family, apoptosis inhibitory protein 5), has been implicated in activation of NLRC4 (refs 7-11), but is widely assumed to have only an auxiliary role, as NAIP5 is often dispensable for NLRC4 activation. However, Naip5 is a member of a small multigene family, raising the possibility of redundancy and functional specialization among Naip genes. Here we show in mice that different NAIP paralogues determine the specificity of the NLRC4 inflammasome for distinct bacterial ligands. In particular, we found that activation of endogenous NLRC4 by bacterial PrgJ requires NAIP2, a previously uncharacterized member of the NAIP gene family, whereas NAIP5 and NAIP6 activate NLRC4 specifically in response to bacterial flagellin. We dissected the biochemical mechanism underlying the requirement for NAIP proteins by use of a reconstituted NLRC4 inflammasome system. We found that NAIP proteins control ligand-dependent oligomerization of NLRC4 and that the NAIP2-NLRC4 complex physically associates with PrgJ but not flagellin, whereas NAIP5-NLRC4 associates with flagellin but not PrgJ. Our results identify NAIPs as immune sensor proteins and provide biochemical evidence for a simple receptor-ligand model for activation of the NAIP-NLRC4 inflammasomes.
© 2011 Macmillan Publishers Limited. All rights reserved
Conflict of interest statement
The authors declare no competing financial interests.
Figures
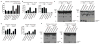
Comment in
-
Inflammasome: NAIPs: pathogen-sensing proteins.Nat Rev Immunol. 2011 Sep 9;11(10):644. doi: 10.1038/nri3069. Nat Rev Immunol. 2011. PMID: 21904388 No abstract available.
-
Immunology: recognition of a unique partner.Nature. 2011 Sep 28;477(7366):543-4. doi: 10.1038/477543a. Nature. 2011. PMID: 21956324 No abstract available.
References
-
- Schroder K, Tschopp J. The inflammasomes. Cell. 2010;140:821–832. - PubMed
-
- Ting JP, Kastner DL, Hoffman HM. CATERPILLERs, pyrin and hereditary immunological disorders. Nat Rev Immunol. 2006;6:183–195. - PubMed
-
- Franchi L, et al. Cytosolic flagellin requires Ipaf for activation of caspase-1 and interleukin 1beta in salmonella-infected macrophages. Nat Immunol. 2006;7:576–582. - PubMed
-
- Miao EA, et al. Cytoplasmic flagellin activates caspase-1 and secretion of interleukin 1beta via Ipaf. Nat Immunol. 2006;7:569–575. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

