Calpain-1 cleaves Rad21 to promote sister chromatid separation
- PMID: 21876002
- PMCID: PMC3209327
- DOI: 10.1128/MCB.06075-11
Calpain-1 cleaves Rad21 to promote sister chromatid separation
Abstract
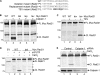
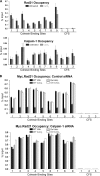
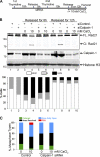
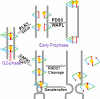
Defining the mechanisms of chromosomal cohesion and dissolution of the cohesin complex from chromatids is important for understanding the chromosomal missegregation seen in many tumor cells. Here we report the identification of a novel cohesin-resolving protease and describe its role in chromosomal segregation. Sister chromatids are held together by cohesin, a multiprotein ring-like complex comprised of Rad21, Smc1, Smc3, and SA2 (or SA1). Cohesin is known to be removed from vertebrate chromosomes by two distinct mechanisms, namely, the prophase and anaphase pathways. First, PLK1-mediated phosphorylation of SA2 in prophase leads to release of cohesin from chromosome arms, leaving behind centromeric cohesins that continue to hold the sisters together. Then, at the onset of anaphase, activated separase cleaves the centromeric cohesin Rad21, thereby opening the cohesin ring and allowing the sister chromatids to separate. We report here that the calcium-dependent cysteine endopeptidase calpain-1 is a Rad21 peptidase and normally localizes to the interphase nuclei and chromatin. Calpain-1 cleaves Rad21 at L192, in a calcium-dependent manner. We further show that Rad21 cleavage by calpain-1 promotes separation of chromosome arms, which coincides with a calcium-induced partial loss of cohesin at several chromosomal loci. Engineered cleavage of Rad21 at the calpain-cleavable site without activation of calpain-1 can lead to a loss of sister chromatid cohesion. Collectively, our work reveals a novel function of calpain-1 and describes an additional pathway for sister chromatid separation in humans.
Figures





Similar articles
-
Characterization of the interaction between the cohesin subunits Rad21 and SA1/2.PLoS One. 2013 Jul 12;8(7):e69458. doi: 10.1371/journal.pone.0069458. Print 2013. PLoS One. 2013. PMID: 23874961 Free PMC article.
-
The hsSsu72 phosphatase is a cohesin-binding protein that regulates the resolution of sister chromatid arm cohesion.EMBO J. 2010 Oct 20;29(20):3544-57. doi: 10.1038/emboj.2010.217. Epub 2010 Sep 3. EMBO J. 2010. PMID: 20818333 Free PMC article.
-
A new role for the mitotic RAD21/SCC1 cohesin in meiotic chromosome cohesion and segregation in the mouse.EMBO Rep. 2004 Apr;5(4):378-84. doi: 10.1038/sj.embor.7400121. Epub 2004 Mar 19. EMBO Rep. 2004. PMID: 15031719 Free PMC article.
-
The expanding phenotypes of cohesinopathies: one ring to rule them all!Cell Cycle. 2019 Nov;18(21):2828-2848. doi: 10.1080/15384101.2019.1658476. Epub 2019 Sep 13. Cell Cycle. 2019. PMID: 31516082 Free PMC article. Review.
-
Sister chromatid resolution: a cohesin releasing network and beyond.Chromosoma. 2010 Oct;119(5):459-67. doi: 10.1007/s00412-010-0271-z. Epub 2010 Mar 30. Chromosoma. 2010. PMID: 20352243 Review.
Cited by
-
Molecular basis of calpain cleavage and inactivation of the sodium-calcium exchanger 1 in heart failure.J Biol Chem. 2014 Dec 5;289(49):33984-98. doi: 10.1074/jbc.M114.602581. Epub 2014 Oct 21. J Biol Chem. 2014. PMID: 25336645 Free PMC article.
-
Cohesin subunit RAD21: From biology to disease.Gene. 2020 Oct 20;758:144966. doi: 10.1016/j.gene.2020.144966. Epub 2020 Jul 17. Gene. 2020. PMID: 32687945 Free PMC article. Review.
-
PKC signaling contributes to chromatin decondensation and is required for competence to respond to IL-2 during T cell activation.Cell Immunol. 2020 Jan;347:104027. doi: 10.1016/j.cellimm.2019.104027. Epub 2019 Dec 11. Cell Immunol. 2020. PMID: 31864664 Free PMC article.
-
Protein Phosphatase 1c Associated with the Cardiac Sodium Calcium Exchanger 1 Regulates Its Activity by Dephosphorylating Serine 68-phosphorylated Phospholemman.J Biol Chem. 2016 Feb 26;291(9):4561-79. doi: 10.1074/jbc.M115.677898. Epub 2015 Dec 14. J Biol Chem. 2016. PMID: 26668322 Free PMC article.
-
Therapeutic relevance of the protein phosphatase 2A in cancer.Oncotarget. 2016 Sep 20;7(38):61544-61561. doi: 10.18632/oncotarget.11399. Oncotarget. 2016. PMID: 27557495 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
