Noninferiority effects on glycemic control and β-cell function improvement in newly diagnosed type 2 diabetes patients: basal insulin monotherapy versus continuous subcutaneous insulin infusion treatment
- PMID: 21877913
- PMCID: PMC3249622
- DOI: 10.1089/dia.2011.0123
Noninferiority effects on glycemic control and β-cell function improvement in newly diagnosed type 2 diabetes patients: basal insulin monotherapy versus continuous subcutaneous insulin infusion treatment
Erratum in
- Diabetes Technol Ther. 2014 Mar;16(3):193
Abstract
Aims: In newly diagnosed type 2 diabetes mellitus (T2DM) patients, short-term insulin therapy might improve β-cell function and glycemic control. This study aimed to compare the effects of basal insulin monotherapy with continuous subcutaneous insulin infusion (CSII) treatment.
Methods: Fifty-nine cases of newly diagnosed T2DM patients with fasting plasma glucose of 9.0-16.7 mmol/L were recruited into this study. They were hospitalized and randomly assigned to a basal insulin monotherapy group (n=27) or a CSII group (n=32). Insulin dosage was titrated according to fasting capillary blood glucose levels, and treatment was stopped after 2 weeks. Intravenous glucose tolerance tests were performed, and blood glucose, insulin, C-peptide, and lipid profiles were measured before therapy and 2 days after therapy withdrawal.
Results: Both treatments reduced fasting and postprandial blood glucose levels (after treatment vs. baseline, both P<0.05). Fasting glycemic control target was achieved in 52 cases (88.14%) with 2 weeks of insulin treatment, and there were no significant differences between the glargine and CSII groups (P=0.059). The time to achieve fasting glycemic target in the CSII group was shorter than that in the glargine group (P<0.01). Plasma lipid profiles such as triglycerides and total cholesterol also decreased significantly after the intervention. Overall β-cell function improved significantly after insulin intervention (P<0.01). Variation did not differ between two groups, nor did the effects on insulin and C-peptide secretion (P>0.05).
Conclusions: The effect of basal insulin monotherapy was similar to that of CSII, and thus basal insulin monotherapy might be a reasonable alternative to CSII for initial insulin therapy in newly diagnosed T2DM patients.
Figures
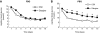
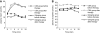

Similar articles
-
A Randomized Trial of Insulin Glargine plus Oral Hypoglycemic Agents versus Continuous Subcutaneous Insulin Infusion to Treat Newly Diagnosed Type 2 Diabetes.J Diabetes Res. 2018 Oct 21;2018:2791584. doi: 10.1155/2018/2791584. eCollection 2018. J Diabetes Res. 2018. PMID: 30420969 Free PMC article. Clinical Trial.
-
Insulin requirement profiles and related factors of insulin pump therapy in patients with type 2 diabetes.Sci China Life Sci. 2019 Nov;62(11):1506-1513. doi: 10.1007/s11427-018-9530-3. Epub 2019 May 22. Sci China Life Sci. 2019. PMID: 31197759
-
Comparison of the Effects of Continuous Subcutaneous Insulin Infusion and Add-On Therapy with Sitagliptin in Patients with Newly Diagnosed Type 2 Diabetes Mellitus.J Diabetes Res. 2016;2016:9849328. doi: 10.1155/2016/9849328. Epub 2015 Dec 21. J Diabetes Res. 2016. PMID: 26798658 Free PMC article. Clinical Trial.
-
Impact of improving postprandial glycemic control with intensifying insulin therapy in type 2 diabetes.Postgrad Med. 2017 Nov;129(8):791-800. doi: 10.1080/00325481.2017.1389601. Epub 2017 Oct 14. Postgrad Med. 2017. PMID: 29032696 Review.
-
Long-acting insulin analogs versus insulin pump therapy for the treatment of type 1 and type 2 diabetes.Diabetes Care. 2008 Feb;31 Suppl 2:S140-5. doi: 10.2337/dc08-s235. Diabetes Care. 2008. PMID: 18227475 Review.
Cited by
-
Role of Adaptive and Innate Immunity in Type 2 Diabetes Mellitus.J Diabetes Res. 2018 Nov 8;2018:7457269. doi: 10.1155/2018/7457269. eCollection 2018. J Diabetes Res. 2018. PMID: 30533447 Free PMC article. Review.
-
Apical periodontitis promotes insulin resistance and alters adaptive immunity markers in rats.Saudi Dent J. 2021 Dec;33(8):979-986. doi: 10.1016/j.sdentj.2021.07.005. Epub 2021 Jul 14. Saudi Dent J. 2021. PMID: 34916766 Free PMC article.
-
Early Treatment with Basal Insulin Glargine in People with Type 2 Diabetes: Lessons from ORIGIN and Other Cardiovascular Trials.Diabetes Ther. 2016 Jun;7(2):187-201. doi: 10.1007/s13300-016-0153-3. Epub 2016 Feb 10. Diabetes Ther. 2016. PMID: 26861811 Free PMC article. Review.
-
Insulin glargine effectively achieves glycemic control and improves insulin resistance in patients with early type 2 diabetes that exhibit a high risk for cardiovascular disease.Exp Ther Med. 2014 Jul;8(1):147-152. doi: 10.3892/etm.2014.1688. Epub 2014 Apr 24. Exp Ther Med. 2014. PMID: 24944613 Free PMC article.
-
Physiological and Immunological Causes of the Susceptibility of Chronic Inflammatory Patients to COVID-19 Infection: Focus on Diabetes.Front Endocrinol (Lausanne). 2021 Mar 4;12:576412. doi: 10.3389/fendo.2021.576412. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 33746897 Free PMC article. Review.
References
-
- Reaven GM. HOMA-beta in the UKPDS and ADOPT. Is the natural history of type 2 diabetes characterized by a progressive and inexorable loss of insulin secretory function? Maybe? Maybe not? Diabtes Vasc Dis Res. 2009;6:133–138. - PubMed
-
- Wajchenberg BL. Clinical approaches to preserve beta-cell function in diabetes. Adv Exp Med Biol. 2010;654:515–535. - PubMed
-
- Wajchenberg BL. β-Cell failure in diabetes and preservation by clinical treatment. Endocr Rev. 2007;28:187–218. - PubMed
-
- Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS33) UK Prospective Diabetes Study (UKPDS) Group. Lancet. 1998;52:837–853. Erratum in Lancet 1999;354:602. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

