Regulation of sororin by Cdk1-mediated phosphorylation
- PMID: 21878504
- PMCID: PMC3166038
- DOI: 10.1242/jcs.085431
Regulation of sororin by Cdk1-mediated phosphorylation
Abstract
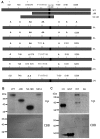
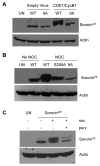
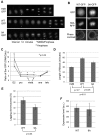
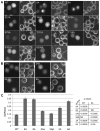
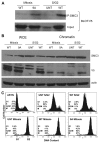
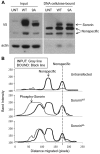
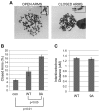
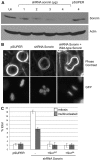
Tumor cells are commonly aneuploid, a condition contributing to cancer progression and drug resistance. Understanding how chromatids are linked and separated at the appropriate time will help uncover the basis of aneuploidy and will shed light on the behavior of tumor cells. Cohesion of sister chromatids is maintained by the multi-protein complex cohesin, consisting of Smc1, Smc3, Scc1 and Scc3. Sororin associates with the cohesin complex and regulates the segregation of sister chromatids. Sororin is phosphorylated in mitosis; however, the role of this modification is unclear. Here we show that mutation of potential cyclin-dependent kinase 1 (Cdk1) phosphorylation sites leaves sororin stranded on chromosomes and bound to cohesin throughout mitosis. Sororin can be precipitated from cell lysates with DNA-cellulose, and only the hypophosphorylated form of sororin shows this association. These results suggest that phosphorylation of sororin causes its release from chromatin in mitosis. Also, the hypophosphorylated form of sororin increases cohesion between sister chromatids, suggesting that phosphorylation of sororin by Cdk1 influences sister chromatid cohesion. Finally, phosphorylation-deficient sororin can alleviate the mitotic block that occurs upon knockdown of endogenous sororin. This mitotic block is abolished by ZM447439, an Aurora kinase inhibitor, suggesting that prematurely separated sister chromatids activate the spindle assembly checkpoint through an Aurora kinase-dependent pathway.
Figures
Similar articles
-
Phosphorylation-enabled binding of SGO1-PP2A to cohesin protects sororin and centromeric cohesion during mitosis.Nat Cell Biol. 2013 Jan;15(1):40-9. doi: 10.1038/ncb2637. Epub 2012 Dec 16. Nat Cell Biol. 2013. PMID: 23242214 Free PMC article.
-
Sororin is a master regulator of sister chromatid cohesion and separation.Cell Cycle. 2012 Jun 1;11(11):2073-83. doi: 10.4161/cc.20241. Epub 2012 Jun 1. Cell Cycle. 2012. PMID: 22580470 Free PMC article. Review.
-
Multiple Levels of Regulation of Sororin by Cdk1 and Aurora B.J Cell Biochem. 2016 Feb;117(2):351-60. doi: 10.1002/jcb.25277. J Cell Biochem. 2016. PMID: 26177583 Free PMC article.
-
C-terminus of Sororin interacts with SA2 and regulates sister chromatid cohesion.Cell Cycle. 2015;14(6):820-6. doi: 10.1080/15384101.2014.1000206. Cell Cycle. 2015. PMID: 25608232 Free PMC article.
-
Sister chromatid cohesion.Cold Spring Harb Perspect Biol. 2012 Nov 1;4(11):a011130. doi: 10.1101/cshperspect.a011130. Cold Spring Harb Perspect Biol. 2012. PMID: 23043155 Free PMC article. Review.
Cited by
-
Cyclin-Dependent Kinase 1 (CDK1) is Co-Expressed with CDCA5: Their Functions in Gastric Cancer Cell Line MGC-803.Med Sci Monit. 2020 Aug 6;26:e923664. doi: 10.12659/MSM.923664. Med Sci Monit. 2020. PMID: 32759885 Free PMC article.
-
Functioning mechanisms of Shugoshin-1 in centromeric cohesion during mitosis.Essays Biochem. 2020 Sep 4;64(2):289-297. doi: 10.1042/EBC20190077. Essays Biochem. 2020. PMID: 32451529 Free PMC article. Review.
-
A kinase-dependent role for Haspin in antagonizing Wapl and protecting mitotic centromere cohesion.EMBO Rep. 2018 Jan;19(1):43-56. doi: 10.15252/embr.201744737. Epub 2017 Nov 14. EMBO Rep. 2018. PMID: 29138236 Free PMC article.
-
Targeting CDK1 in cancer: mechanisms and implications.NPJ Precis Oncol. 2023 Jun 13;7(1):58. doi: 10.1038/s41698-023-00407-7. NPJ Precis Oncol. 2023. PMID: 37311884 Free PMC article. Review.
-
The Functions and Mechanisms of the Cohesin Complex in Regulating the Fate Determinations of Stem Cells.Research (Wash D C). 2025 Jul 10;8:0757. doi: 10.34133/research.0757. eCollection 2025. Research (Wash D C). 2025. PMID: 40642059 Free PMC article. Review.
References
-
- Beausoleil S. A., Villen J., Gerber S. A., Rush J., Gygi S. P. (2006). A probability-based approach for high-throughput protein phosphorylation analysis and site localization. Nat. Biotechnol. 24, 1285-1292 - PubMed
-
- Cantin G. T., Yi W., Lu B., Park S. K., Xu T., Lee J. D., Yates J. R., 3rd (2008). Combining protein-based IMAC, peptide-based IMAC, and MudPIT for efficient phosphoproteomic analysis. J. Proteome Res. 7, 1346-1351 - PubMed
-
- Diaz-Martinez L. A., Gimenez-Abian J. F., Clarke D. J. (2007). Regulation of centromeric cohesion by sororin independently of the APC/C. Cell Cycle 6, 714-724 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

