Uroporphyria in the Cyp1a2-/- mouse
- PMID: 21880518
- PMCID: PMC3223295
- DOI: 10.1016/j.bcmd.2011.07.006
Uroporphyria in the Cyp1a2-/- mouse
Abstract
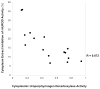
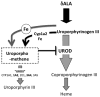
Cytochrome P4501A2 (Cyp1a2) is important in the development of uroporphyria in mice, a model of porphyria cutanea tarda in humans. Heretofore, mice homozygous for the Cyp1a2-/- mutation do not develop uroporphyria with treatment regimens that result in uroporphyria in wild-type mice. Here we report uroporphyria development in Cyp1a2-/- mice additionally null for both alleles of the hemochromatosis (Hfe) gene and heterozygous for deletion of the uroporphyrinogen decarboxylase (Urod) gene (genotype: Cyp1a2-/-;Hfe-/-;Urod+/-), demonstrating that upon adding porphyria-predisposing genetic manipulations, Cyp1a2 is not essential. Cyp1a2-/-;Hfe-/-;Urod+/- mice were treated with various combinations of an iron-enriched diet, parenteral iron-dextran, drinking water containing δ-aminolevulinic acid and intraperitoneal Aroclor 1254 (a polychlorinated biphenyl mixture) and analyzed for uroporphyrin accumulation. Animals fed an iron-enriched diet alone did not develop uroporphyria but uroporphyria developed with all treatments that included iron supplementation and δ-aminolevulinic acid, even with a regimen without Aroclor 1254. Hepatic porphyrin levels correlated with low UROD activity and high levels of an inhibitor of UROD but marked variability in the magnitude of the porphyric response was present in all treatment groups. Gene expression profiling revealed no major differences between genetically identical triple cross mice exhibiting high and low magnitude porphyric responses from iron-enriched diet and iron-dextran supplementation, and δ-aminolevulinic acid. Even though the variation in porphyric response did not parallel the hepatic iron concentration, the results are compatible with the presence of a Cyp1a2-independent, iron-dependent pathway for the generation of uroporphomethene, the UROD inhibitor required for the expression of uroporphyria in mice and PCT in humans.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures
Similar articles
-
Uroporphyria in mice: thresholds for hepatic CYP1A2 and iron.Hepatology. 2002 Apr;35(4):912-21. doi: 10.1053/jhep.2002.32487. Hepatology. 2002. PMID: 11915039
-
Uroporphyria in Hfe mutant mice given 5-aminolevulinate: a new model of Fe-mediated porphyria cutanea tarda.Hepatology. 2001 Feb;33(2):406-12. doi: 10.1053/jhep.2001.21409. Hepatology. 2001. PMID: 11172342
-
Uroporphyria in the uroporphyrinogen decarboxylase-deficient mouse: Interplay with siderosis and polychlorinated biphenyl exposure.Hepatology. 2002 Oct;36(4 Pt 1):805-11. doi: 10.1053/jhep.2002.35621. Hepatology. 2002. PMID: 12297827
-
Complex gene-chemical interactions: hepatic uroporphyria as a paradigm.Chem Res Toxicol. 2010 Apr 19;23(4):712-23. doi: 10.1021/tx900298k. Chem Res Toxicol. 2010. PMID: 20099833 Review.
-
[Pathogenesis of porphyria cutanea tarda].Orv Hetil. 2000 Apr 2;141(14):709-13. Orv Hetil. 2000. PMID: 10803012 Review. Hungarian.
Cited by
-
A mouse model of hereditary coproporphyria identified in an ENU mutagenesis screen.Dis Model Mech. 2017 Aug 1;10(8):1005-1013. doi: 10.1242/dmm.029116. Epub 2017 Jun 9. Dis Model Mech. 2017. PMID: 28600349 Free PMC article.
-
Porphyria cutanea tarda: a unique iron-related disorder.Hematology Am Soc Hematol Educ Program. 2024 Dec 6;2024(1):450-456. doi: 10.1182/hematology.2024000664. Hematology Am Soc Hematol Educ Program. 2024. PMID: 39644053 Free PMC article. Review.
-
HFE gene: Structure, function, mutations, and associated iron abnormalities.Gene. 2015 Dec 15;574(2):179-92. doi: 10.1016/j.gene.2015.10.009. Epub 2015 Oct 9. Gene. 2015. PMID: 26456104 Free PMC article. Review.
-
New insights of CYP1A in endogenous metabolism: a focus on single nucleotide polymorphisms and diseases.Acta Pharm Sin B. 2020 Jan;10(1):91-104. doi: 10.1016/j.apsb.2019.11.016. Epub 2019 Dec 2. Acta Pharm Sin B. 2020. PMID: 31998606 Free PMC article. Review.
-
Iron in Porphyrias: Friend or Foe?Diagnostics (Basel). 2022 Jan 21;12(2):272. doi: 10.3390/diagnostics12020272. Diagnostics (Basel). 2022. PMID: 35204362 Free PMC article. Review.
References
-
- Phillips JD, Kushner JP. The Porphyrias. In: Nathan DG, editor. Nathan and Oski’s Hematology of Infancy and Childhood. Saunders; Philadelphia: 2009. pp. 571–612.
-
- Cam C, Nigogosyan G. Acquired Toxic Porphyria Cutanea Tarda due to Hexachlorobenzene. JAMA. 1963;183:90–93. - PubMed
-
- Franklin MR, Phillips JD, Kushner JP. Attenuation of polychlorinated biphenyl induced uroporphyria by iron deprivation. Env Toxicol Pharmacol. 2005;20:417–423. - PubMed
-
- Gorman N, Ross KL, Walton HS, et al. Uroporphyria in mice: thresholds for hepatic CYP1A2 and iron. Hepatology. 2002;35:912–21. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

