The atypical Guanine-nucleotide exchange factor, dock7, negatively regulates schwann cell differentiation and myelination
- PMID: 21880919
- PMCID: PMC6703263
- DOI: 10.1523/JNEUROSCI.2738-11.2011
The atypical Guanine-nucleotide exchange factor, dock7, negatively regulates schwann cell differentiation and myelination
Abstract
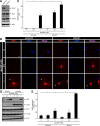
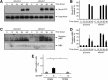
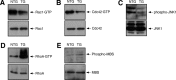
In development of the peripheral nervous system, Schwann cells proliferate, migrate, and ultimately differentiate to form myelin sheath. In all of the myelination stages, Schwann cells continuously undergo morphological changes; however, little is known about their underlying molecular mechanisms. We previously cloned the dock7 gene encoding the atypical Rho family guanine-nucleotide exchange factor (GEF) and reported the positive role of Dock7, the target Rho GTPases Rac/Cdc42, and the downstream c-Jun N-terminal kinase in Schwann cell migration (Yamauchi et al., 2008). We investigated the role of Dock7 in Schwann cell differentiation and myelination. Knockdown of Dock7 by the specific small interfering (si)RNA in primary Schwann cells promotes dibutyryl cAMP-induced morphological differentiation, indicating the negative role of Dock7 in Schwann cell differentiation. It also results in a shorter duration of activation of Rac/Cdc42 and JNK, which is the negative regulator of myelination, and the earlier activation of Rho and Rho-kinase, which is the positive regulator of myelination. To obtain the in vivo evidence, we generated Dock7 short hairpin (sh)RNA transgenic mice. They exhibited a decreased expression of Dock7 in the sciatic nerves and enhanced myelin thickness, consistent with in vitro observation. The effects of the in vivo knockdown on the signals to Rho GTPases are similar to those of the in vitro knockdown. Collectively, the signaling through Dock7 negatively regulates Schwann cell differentiation and the onset of myelination, demonstrating the unexpected role of Dock7 in the interplay between Schwann cell migration and myelination.
Figures









References
-
- Bar-Sagi D, Hall A. Ras and Rho GTPases: a family reunion. Cell. 2000;103:227–238. - PubMed
-
- Benninger Y, Thurnherr T, Pereira JA, Krause S, Wu X, Chrostek-Grashoff A, Herzog D, Nave KA, Franklin RJ, Meijer D, Brakebusch C, Suter U, Relvas JB. Essential and distinct roles for Cdc42 and Rac1 in the regulation of Schwann cell biology during peripheral nervous system development. J Cell Biol. 2007;177:1051–1061. - PMC - PubMed
-
- Bunge RP. Expanding roles for the Schwann cell: ensheathment, myelination, trophism and regeneration. Curr Opin Neurobiol. 1993;3:805–809. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
