Night-time symptoms: a forgotten dimension of COPD
- PMID: 21881146
- PMCID: PMC9584119
- DOI: 10.1183/09059180.00004311
Night-time symptoms: a forgotten dimension of COPD
Abstract
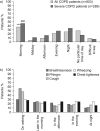
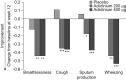
Sleep quality is often poor in patients with chronic obstructive pulmonary disease (COPD), but these night-time symptoms are frequently unnoticed by physicians and/or not reported by patients themselves. Therefore, the prevalence and clinical impact of sleep disturbances and night-time symptoms in COPD is not well understood and has not been a clinical focus to date. To address this gap, an expert panel meeting was convened in Barcelona, Spain, in March 2011 to discuss the aetiology, evolution, burden, long-term clinical consequences and optimal management of night-time symptoms in COPD. The term "night-time symptoms" in COPD has not been distinctly defined in an objective sense but epidemiological data suggests that the prevalence of nocturnal symptoms and symptomatic sleep disturbance may exceed 75% in patients with COPD. The panel concluded that night-time symptoms in COPD are prevalent and bothersome; that their cause(s) are multiple and include demographic factors, such as age and obesity, pharmacotherapy, disease-specific symptoms and the presence of comorbid sleep disorders, and other medical conditions; and that potential long-term consequences can include lung function changes, increased exacerbation frequency, emergence or worsening of cardiovascular disease, cognitive effects, depression, impaired quality of life and increased mortality. To date, few interventional studies have investigated them, but emerging data suggest that bronchodilator therapy can improve them if deployed appropriately. In summary, night-time symptoms in COPD warrant further clinical investigation with validated tools.
Conflict of interest statement
This study was supported by an unrestricted educational grant from Almirall S.A., Barcelona, Spain. A. Agusti has received honoraria for participating in advisory boards, lecturing at meetings and/or funds for research in the field of COPD from Almirall, AstraZeneca, Boehringer Ingelheim, Chiesi, Esteve, GSK, MSD, Novartis, Nycomed, Pfizer, Roche, and Procter and Gamble. J. Hedner has received fees for speaking at research symposia by Weinmann GmbH and a fee for consulting with Almirall S.A. He has received two unrestricted grants from ResMed Inc. and Philips Respironics for maintenance of the European Sleep Apnea Database (ESADA). J.M. Marin has received fees for speaking (less than €1,000), and has received public funds for research from the Institut Carlos III, Madrid, Spain. M. Cazzola has received financial support for research and attending meetings and fees for speaking and consulting from Abbott, Almirall, AstraZeneca, Boehringer Ingelheim, Chiesi Farmaceutici, Dey, Eli Lilly, Gentili, GSK, Lallemand Pharma, Meda, Menarini Farmaceutici, Novartis, Nycomed, Pfizer, Sanovel, Sanofi-Aventis, Sigma-Tau and Valeas. S. Rennard has served as a consultant or participated in advisory boards for: ABIM, Able Associates, Adelphi Research, Almirall, APT, Aradigm, Argenta, AstraZeneca, BI (ACCP), Biostrategies, BoomCom, Britnall and Nicolini, Capital Research, Chiesi, Clinical Advisors, CommonHealth, Complete Medical Group, Consult Complete, COPDForum, DataMonitor, Decision Resources, Defined Health, Dey, Dunn Group, Easton Associates, Enterprise Analysis, Equinox, Forest, Fulcrum, Gerson Lehman, GSK, Guidepoint, Hoffman LaRoche, IMS, Informed, Inspire, Insyght, KOL Connection, Leerink Swan, M. Pankove, MDRx Financial, MedaCorp, Medimmune, Mpex, Novartis, Nycomed, Oriel, Otsuka, Pearl, Pennside Partners, Pfizer, Pharma Ventures, Pharmaxis, Pick Research, Prescott, Price Waterhouse, Propagate, Pulmatrix, Pulmonary Reviews, Quadrant, Reckner Associates, Recruiting Resource, Reviews and Trends in COPD/Convergent Health Solutions, Roche, Sacoor, Schering, Schlesinger Medical, Scimed, Smith Research, Sudler and Hennessey, Talecris, Theravance, UBC and Uptake Medical, Vantage Point; has received lecture fees from AAAAI, Am Col Osteopathic Physicians, Asan Medical Center, ATS, AstraZeneca, California Soc Allergy, Convergent Health Solutions for Reviews and Trends in COPD, COPDFoundation, Creative Educational Concepts, Dey, Duke, France Foundation, Information TV, University of California-Los Angeles, Network for Continuing Education, Novartis, Nycomed, Otsuka, Pfizer, Sarasota Mem Hospital, Spanish Thoracic Society, University of Washington, University of Alabama-Birmingham, University of Pittsburgh, University of British Columbia, University of California-Davis and VA Sioux Falls; and has received industry sponsored grants from AstraZeneca, Biomarck, Centocor, GlaxoSmithKline, Mpex, Nabi, Novartis, Otsuka and Pfizer. S. Rennard has also received tobacco industry funding, specifically from the tobacco industry for studies relating to harm reduction and to the impact of tobacco smoke on stem cells, and has also consulted with RJ Reynolds, without personal fee, on the topic of harm reduction. He received funding from RJ Reynolds to evaluate the effect of a harm reduction product in normal smokers (1996) and in subjects with chronic bronchitis (1999) and to assess the effect of smoking cessation on lower respiratory tract inflammation (2000); participated in a Philip Morris multicentre study to assess biomarkers of smoke exposure (2002); received funding for a clinical trial from the Institute for Science and Health (2005), which receives support from the tobacco industry, to evaluate biomarkers in exhaled breath associated with smoking cessation and reduction (this study was supplemented with funding from Lorillard and RJ Reynolds); received a grant from the Philip Morris External Research Program (2005) to assess the impact of cigarette smoking on circulating stem cells in the mouse. He has consulted with RJ Reynolds on the topic of harm reduction until 2007, but did not receive personal remuneration for this. S. Rennard is not currently involved in tobacco-industry funded projects; all ties with tobacco industry companies and entities supported by tobacco companies were terminated in 2007.
Figures


References
-
- American Thoracic Society, European Respiratory Society. Standards for the Diagnosis and Management of Patients with COPD. New York, American Thoracic Society; Lausanne, European Respiratory Society, 2004.
-
- Parish JM. Sleep-related problems in common medical conditions. Chest 2009; 135: 563–572. - PubMed
-
- Global Initiative for Chronic Obstructive Lung Disease. Global Strategy for Diagnosis, Management, and Prevention of COPD. Global Initiative for Chronic Obstructive Lung Disease, 2010.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
