Smad4 deficiency in T cells leads to the Th17-associated development of premalignant gastroduodenal lesions in mice
- PMID: 21881210
- PMCID: PMC3195457
- DOI: 10.1172/JCI45114
Smad4 deficiency in T cells leads to the Th17-associated development of premalignant gastroduodenal lesions in mice
Abstract
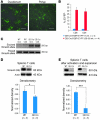
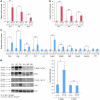
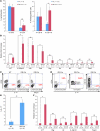
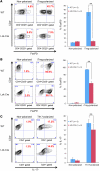
While there is evidence that specific T cell populations can promote the growth of established tumors, instances where T cell activity causes neoplasms to arise de novo are infrequent. Here, we employed two conditional mutagenesis systems to delete the TGF-β signaling pathway component Smad4 in T cells and observed the spontaneous development of massive polyps within the gastroduodenal regions of mice. The epithelial lesions contained increased levels of transcripts encoding IL-11, IL-6, TGF-β, IL-1β, and TNF-α, and lamina propria cells isolated from lesions contained abundant IL-17A+CD4+ T cells. Furthermore, we found that Smad4 deficiency attenuated TGF-β-mediated in vitro polarization of FoxP3+CD4+ T cells, but not IL-17A+CD4+ T cells, suggesting that the epithelial lesions may have arisen as a consequence of unchecked Th17 cell activity. Proinflammatory cytokine production likely accounted for the raised levels of IL-11, a cytokine known to promote gastric epithelial cell survival and hyperplasia. Consistent with IL-11 having a pathogenic role in this model, we found evidence of Stat3 activation in the gastric polyps. Thus, our data indicate that a chronic increase in gut Th17 cell activity can be associated with the development of premalignant lesions of the gastroduodenal region.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

