Everolimus is an active agent in medullary thyroid cancer: a clinical and in vitro study
- PMID: 21883896
- PMCID: PMC3823224
- DOI: 10.1111/j.1582-4934.2011.01438.x
Everolimus is an active agent in medullary thyroid cancer: a clinical and in vitro study
Abstract
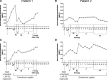
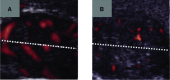
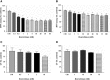
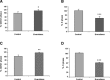
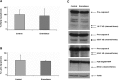
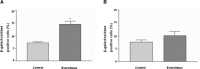
Everolimus, an mTOR inhibitor, which has been demonstrated to induce anti-tumour effects in different types of neuroendocrine tumours, has never been evaluated in patients with medullary thyroid cancer (MTC). The aim of this study was to evaluate the in vitro and in vivo effects of everolimus in combination with octreotide in MTC. Two patients with progressive metastatic MTC and high calcitonin levels were treated with everolimus 5-10 mg/day. Both patients were under treatment with octreotide LAR at the study entry. An in vitro study was also performed to assess everolimus effects on MTC cell lines (TT and MZ-CRC-1 cells). A tumour response was observed in both patients. Serum calcitonin decreased by 86% in patient 1 and by 42% in patient 2. In TT and MZ-CRC-1 cells, everolimus induced a significant dose-dependent inhibition in cell proliferation. This effect seems to be related to a cell cycle arrest in G(0) /G(1) phase in both cell lines and to the induction of cellular senescence in TT cells. Everolimus in combination with octreotide may be active as anti-tumour therapy in patients with progressive metastatic MTC, suggesting to further evaluate this agent in MTC patients in a large prospective study.
© 2011 The Authors Journal of Cellular and Molecular Medicine © 2011 Foundation for Cellular and Molecular Medicine/Blackwell Publishing Ltd.
Figures
Similar articles
-
The antiproliferative effect of pasireotide LAR alone and in combination with everolimus in patients with medullary thyroid cancer: a single-center, open-label, phase II, proof-of-concept study.Endocrine. 2018 Oct;62(1):46-56. doi: 10.1007/s12020-018-1583-7. Epub 2018 Mar 23. Endocrine. 2018. PMID: 29572709
-
Sorafenib and Mek inhibition is synergistic in medullary thyroid carcinoma in vitro.Endocr Relat Cancer. 2012 Jan 9;19(1):29-38. doi: 10.1530/ERC-11-0155. Print 2012 Feb. Endocr Relat Cancer. 2012. PMID: 22109971 Free PMC article.
-
The rapamycin-derivative RAD001 (everolimus) inhibits cell viability and interacts with the Akt-mTOR-p70S6K pathway in human medullary thyroid carcinoma cells.Mol Cell Endocrinol. 2010 Feb 5;315(1-2):87-94. doi: 10.1016/j.mce.2009.09.027. Epub 2009 Oct 6. Mol Cell Endocrinol. 2010. PMID: 19815051
-
[Calcitonin determination for early diagnosis of medullary thyroid cancer].Chirurg. 2010 Jul;81(7):620, 622-6. doi: 10.1007/s00104-009-1883-9. Chirurg. 2010. PMID: 20549176 Review. German.
-
Clinical review: Incidentally discovered medullary thyroid cancer: diagnostic strategies and treatment.J Clin Endocrinol Metab. 2011 May;96(5):1237-45. doi: 10.1210/jc.2010-2359. Epub 2011 Feb 23. J Clin Endocrinol Metab. 2011. PMID: 21346073 Free PMC article. Review.
Cited by
-
p53 suppresses carcinoma progression by inhibiting mTOR pathway activation.Oncogene. 2015 Jan 29;34(5):589-99. doi: 10.1038/onc.2013.589. Epub 2014 Jan 27. Oncogene. 2015. PMID: 24469052 Free PMC article.
-
The antiproliferative effect of pasireotide LAR alone and in combination with everolimus in patients with medullary thyroid cancer: a single-center, open-label, phase II, proof-of-concept study.Endocrine. 2018 Oct;62(1):46-56. doi: 10.1007/s12020-018-1583-7. Epub 2018 Mar 23. Endocrine. 2018. PMID: 29572709
-
CXCR4/CXCL12/CXCR7 axis is functional in neuroendocrine tumors and signals on mTOR.Oncotarget. 2016 Apr 5;7(14):18865-75. doi: 10.18632/oncotarget.7738. Oncotarget. 2016. PMID: 26934559 Free PMC article.
-
The dual targeting of insulin and insulin-like growth factor 1 receptor enhances the mTOR inhibitor-mediated antitumor efficacy in hepatocellular carcinoma.Oncotarget. 2016 Mar 1;7(9):9718-31. doi: 10.18632/oncotarget.6836. Oncotarget. 2016. PMID: 26756219 Free PMC article.
-
Antagonistic effects of chloroquine on autophagy occurrence potentiate the anticancer effects of everolimus on renal cancer cells.Cancer Biol Ther. 2015;16(4):567-79. doi: 10.1080/15384047.2015.1018494. Epub 2015 Apr 11. Cancer Biol Ther. 2015. PMID: 25866016 Free PMC article.
References
-
- Leboulleux S, Baudin E, Travagli JP, et al. Medullary thyroid carcinoma. Clin Endocrinol. 2004;61:299–310. - PubMed
-
- Sippel RS, Kunnimalaiyaan M, Chen H. Current management of medullary thyroid cancer. Oncologist. 2008;13:539–47. - PubMed
-
- Papotti M, Kumar U, Volante M, et al. Immunohistochemical detection of somatostatin receptor types 1–5 in medullary carcinoma of the thyroid. Clin Endocrinol. 2001;54:641–9. - PubMed
-
- Zatelli MC, Piccin D, Tagliati F, et al. Selective activation of somatostatin receptor subtypes differentially modulates secretion and viability in human medullary thyroid carcinoma primary cultures: potential clinical perspectives. J Clin Endocrinol Metab. 2006;91:2218–24. - PubMed
-
- Lupoli G, Cascone E, Arlotta F, et al. Treatment of advanced medullary thyroid carcinoma with a combination of recombinant interferon alpha-2b and octreotide. Cancer. 1996;78:1114–8. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

