Role of sialidase in glycoprotein utilization by Tannerella forsythia
- PMID: 21885482
- PMCID: PMC3352272
- DOI: 10.1099/mic.0.052498-0
Role of sialidase in glycoprotein utilization by Tannerella forsythia
Abstract
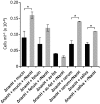
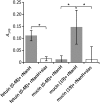
The major bacterial pathogens associated with periodontitis include Tannerella forsythia. We previously discovered that sialic acid stimulates biofilm growth of T. forsythia, and that sialidase activity is key to utilization of sialoconjugate sugars and is involved in host-pathogen interactions in vitro. The aim of this work was to assess the influence of the NanH sialidase on initial biofilm adhesion and growth in experiments where the only source of sialic acid was sialoglycoproteins or human oral secretions. After showing that T. forsythia can utilize sialoglycoproteins for biofilm growth, we showed that growth and initial adhesion with sialylated mucin and fetuin were inhibited two- to threefold by the sialidase inhibitor oseltamivir. A similar reduction (three- to fourfold) was observed with a nanH mutant compared with the wild-type. Importantly, these data were replicated using clinically relevant serum and saliva samples as substrates. In addition, the ability of the nanH mutant to form biofilms on glycoprotein-coated surfaces could be restored by the addition of purified NanH, which we show is able to cleave sialic acid from the model glycoprotein fetuin and, much less efficiently, 9-O-acetylated bovine submaxillary mucin. These data show for the first time that glycoprotein-associated sialic acid is likely to be a key in vivo nutrient source for T. forsythia when growing in a biofilm, and suggest that sialidase inhibitors might be useful adjuncts in periodontal therapy.
Figures
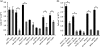


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

