A novel perspective on stem cell homing and mobilization: review on bioactive lipids as potent chemoattractants and cationic peptides as underappreciated modulators of responsiveness to SDF-1 gradients
- PMID: 21886175
- PMCID: PMC5572626
- DOI: 10.1038/leu.2011.242
A novel perspective on stem cell homing and mobilization: review on bioactive lipids as potent chemoattractants and cationic peptides as underappreciated modulators of responsiveness to SDF-1 gradients
Abstract
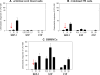
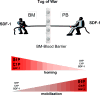
Hematopoietic stem progenitor cells (HSPCs) respond robustly to α-chemokine stromal-derived factor-1 (SDF-1) gradients, and blockage of CXCR4, a seven-transmembrane-spanning G(αI)-protein-coupled SDF-1 receptor, mobilizes HSPCs into peripheral blood. Although the SDF-1-CXCR4 axis has an unquestionably important role in the retention of HSPCs in bone marrow (BM), new evidence shows that, in addition to SDF-1, the migration of HSPCs is directed by gradients of the bioactive lipids sphingosine-1 phosphate and ceramide-1 phosphate. Furthermore, the SDF-1 gradient may be positively primed/modulated by cationic peptides (C3a anaphylatoxin and cathelicidin) and, as previously demonstrated, HSPCs respond robustly even to very low SDF-1 gradients in the presence of priming factors. In this review, we discuss the role of bioactive lipids in stem cell trafficking and the consequences of HSPC priming by cationic peptides. Together, these phenomena support a picture in which the SDF-1-CXCR4 axis modulates homing, BM retention and mobilization of HSPCs in a more complex way than previously envisioned.
Conflict of interest statement
The author declares no conflict of interests.
Figures



References
-
- Levesque JP, Takamatsu Y, Nilsson SK, Haylock DN, Simmons PJ. Vascular cell adhesion molecule-1 (CD106) is cleaved by neutrophil proteases in the bone marrow following hematopoietic progenitor cell mobilization by granulocyte colony-stimulating factor. Blood. 2001;98:1289–1297. - PubMed
-
- Levesque JP, Hendy J, Winkler IG, Takamatsu Y, Simmons PJ. Granulocyte colony-stimulating factor induces the release in the bone marrow of proteases that cleave c-KIT receptor (CD117) from the surface of hematopoietic progenitor cells. Exp Hematol. 2003;31:109–117. - PubMed
-
- Levesque JP, Hendy J, Takamatsu Y, Williams B, Winkler IG, Simmons PJ. Mobilization by either cyclophosphamide or granulocyte colony-stimulating factor transforms the bone marrow into a highly proteolytic environment. Exp Hematol. 2002;30:440–449. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

