Curcumin-loaded apotransferrin nanoparticles provide efficient cellular uptake and effectively inhibit HIV-1 replication in vitro
- PMID: 21887247
- PMCID: PMC3161739
- DOI: 10.1371/journal.pone.0023388
Curcumin-loaded apotransferrin nanoparticles provide efficient cellular uptake and effectively inhibit HIV-1 replication in vitro
Abstract
Background: Curcumin (diferuloylmethane) shows significant activity across a wide spectrum of conditions, but its usefulness is rather limited because of its low bioavailability. Use of nanoparticle formulations to enhance curcumin bioavailability is an emerging area of research.
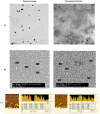
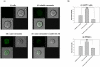
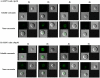
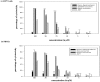
Methodology/principal findings: In the present study, curcumin-loaded apotransferrin nanoparticles (nano-curcumin) prepared by sol-oil chemistry and were characterized by electron and atomic force microscopy. Confocal studies and fluorimetric analysis revealed that these particles enter T cells through transferrin-mediated endocytosis. Nano-curcumin releases significant quantities of drug gradually over a fairly long period, ∼50% of curcumin still remaining at 6 h of time. In contrast, intracellular soluble curcumin (sol-curcumin) reaches a maximum at 2 h followed by its complete elimination by 4 h. While sol-curcumin (GI(50) = 15.6 µM) is twice more toxic than nano-curcumin (GI(50) = 32.5 µM), nano-curcumin (IC(50)<1.75 µM) shows a higher anti-HIV activity compared to sol-curcumin (IC(50) = 5.1 µM). Studies in vitro showed that nano-curcumin prominently inhibited the HIV-1 induced expression of Topo II α, IL-1β and COX-2, an effect not seen with sol-curcumin. Nano-curcumin did not affect the expression of Topoisomerase II β and TNF α. This point out that nano-curcumin affects the HIV-1 induced inflammatory responses through pathways downstream or independent of TNF α. Furthermore, nano-curcumin completely blocks the synthesis of viral cDNA in the gag region suggesting that the nano-curcumin mediated inhibition of HIV-1 replication is targeted to viral cDNA synthesis.
Conclusion: Curcumin-loaded apotransferrin nanoparticles are highly efficacious inhibitors of HIV-1 replication in vitro and promise a high potential for clinical usefulness.
Conflict of interest statement
Figures


Similar articles
-
Topoisomerase II β Gene Specific siRNA Delivery by Nanoparticles Prepared with c-ter Apotransferrin and its Effect on HIV-1 Replication.Mol Biotechnol. 2021 Aug;63(8):732-745. doi: 10.1007/s12033-021-00334-7. Epub 2021 May 16. Mol Biotechnol. 2021. PMID: 33993458
-
An efficient targeted drug delivery through apotransferrin loaded nanoparticles.PLoS One. 2009 Oct 2;4(10):e7240. doi: 10.1371/journal.pone.0007240. PLoS One. 2009. PMID: 19806207 Free PMC article.
-
Regulation of topoisomerase II alpha and beta in HIV-1 infected and uninfected neuroblastoma and astrocytoma cells: involvement of distinct nordihydroguaretic acid sensitive inflammatory pathways.Arch Biochem Biophys. 2007 May 1;461(1):40-9. doi: 10.1016/j.abb.2007.01.026. Epub 2007 Mar 1. Arch Biochem Biophys. 2007. PMID: 17391642
-
The significance of transferrin receptors in oncology: the development of functional nano-based drug delivery systems.Curr Drug Deliv. 2014;11(4):427-43. doi: 10.2174/1567201810666140106115436. Curr Drug Deliv. 2014. PMID: 24387131 Review.
-
HIV-1 associated Topoisomerase IIβ kinase: a potential pharmacological target for viral replication.Curr Pharm Des. 2013;19(26):4776-86. doi: 10.2174/1381612811319260008. Curr Pharm Des. 2013. PMID: 23260019 Review.
Cited by
-
Improved Safety, Bioavailability and Pharmacokinetics of Zidovudine through Lactoferrin Nanoparticles during Oral Administration in Rats.PLoS One. 2015 Oct 13;10(10):e0140399. doi: 10.1371/journal.pone.0140399. eCollection 2015. PLoS One. 2015. PMID: 26461917 Free PMC article.
-
Curcumin ameliorates the permeability of the blood-brain barrier during hypoxia by upregulating heme oxygenase-1 expression in brain microvascular endothelial cells.J Mol Neurosci. 2013 Oct;51(2):344-51. doi: 10.1007/s12031-013-9989-4. Epub 2013 Mar 15. J Mol Neurosci. 2013. PMID: 23494637
-
BRCA1 functions as a novel transcriptional cofactor in HIV-1 infection.Virol J. 2015 Mar 6;12:40. doi: 10.1186/s12985-015-0266-8. Virol J. 2015. PMID: 25879655 Free PMC article.
-
A Comprehensive Review on the Benefits and Problems of Curcumin with Respect to Human Health.Molecules. 2022 Jul 8;27(14):4400. doi: 10.3390/molecules27144400. Molecules. 2022. PMID: 35889273 Free PMC article. Review.
-
Inhibition of HIV-1 by curcumin A, a novel curcumin analog.Drug Des Devel Ther. 2015 Sep 3;9:5051-60. doi: 10.2147/DDDT.S86558. eCollection 2015. Drug Des Devel Ther. 2015. PMID: 26366056 Free PMC article.
References
-
- Sharma OP. Antioxidant activity of curcumin and related compounds. Biochem Pharmacol. 1976;25:1811–1812. - PubMed
-
- Srimal RC, Dhawan BN. Pharmacology of diferuloyl methane (curcumin), a non-steroidal anti-inflammatory agent. J Pharm Pharmacol. 1973;25:447–452. - PubMed
-
- Mahady GB, Pendland SL, Yun G, Lu ZZ. Turmeric (Curcuma longa) and curcumin inhibit the growth of Helicobacter pylori, a group 1 carcinogen. Anticancer Res. 2002;22:4179–4181. - PubMed
-
- Kuttan R, Bhanumathy P, Nirmala K, George MC. Potential anticancer activity of turmeric (Curcuma longa). Cancer Lett. 1985;29:197–202. - PubMed
-
- Kiso Y, Suzuki Y, Watanabe N, Oshima Y, Hikino H. Antihepatotoxic Principles of Curcuma longa Rhizomes1. Planta Medica. 1983;49:185–187. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

