Urothelial plaque formation in post-Golgi compartments
- PMID: 21887288
- PMCID: PMC3161059
- DOI: 10.1371/journal.pone.0023636
Urothelial plaque formation in post-Golgi compartments
Abstract
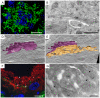
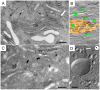
Urothelial plaques are specialized membrane domains in urothelial superficial (umbrella) cells, composed of highly ordered uroplakin particles. We investigated membrane compartments involved in the formation of urothelial plaques in mouse umbrella cells. The Golgi apparatus did not contain uroplakins organized into plaques. In the post-Golgi region, three distinct membrane compartments containing uroplakins were characterized: i) Small rounded vesicles, located close to the Golgi apparatus, were labelled weakly with anti-uroplakin antibodies and they possessed no plaques; we termed them "uroplakin-positive transporting vesicles" (UPTVs). ii) Spherical-to-flattened vesicles, termed "immature fusiform vesicles" (iFVs), were uroplakin-positive in their central regions and contained small urothelial plaques. iii) Flattened "mature fusiform vesicles" (mFVs) contained large plaques, which were densely labelled with anti-uroplakin antibodies. Endoytotic marker horseradish peroxidase was not found in these post-Golgi compartments. We propose a detailed model of de novo urothelial plaque formation in post-Golgi compartments: UPTVs carrying individual 16-nm particles detach from the Golgi apparatus and subsequently fuse into iFV. Concentration of 16-nm particles into plaques and removal of uroplakin-negative membranes takes place in iFVs. With additional fusions and buddings, iFVs mature into mFVs, each carrying two urothelial plaques toward the apical surface of the umbrella cell.
Conflict of interest statement
Figures



Similar articles
-
MAL facilitates the incorporation of exocytic uroplakin-delivering vesicles into the apical membrane of urothelial umbrella cells.Mol Biol Cell. 2012 Apr;23(7):1354-66. doi: 10.1091/mbc.E11-09-0823. Epub 2012 Feb 9. Mol Biol Cell. 2012. PMID: 22323295 Free PMC article.
-
SNX31: a novel sorting nexin associated with the uroplakin-degrading multivesicular bodies in terminally differentiated urothelial cells.PLoS One. 2014 Jun 10;9(6):e99644. doi: 10.1371/journal.pone.0099644. eCollection 2014. PLoS One. 2014. PMID: 24914955 Free PMC article.
-
Expression and localization of four uroplakins in urothelial preneoplastic lesions.Histochem Cell Biol. 2011 Oct;136(4):491-500. doi: 10.1007/s00418-011-0857-4. Epub 2011 Aug 19. Histochem Cell Biol. 2011. PMID: 21853341
-
The Golgi complex: An organelle that determines urothelial cell biology in health and disease.Histochem Cell Biol. 2022 Sep;158(3):229-240. doi: 10.1007/s00418-022-02121-0. Epub 2022 Jun 30. Histochem Cell Biol. 2022. PMID: 35773494 Free PMC article. Review.
-
Formation of asymmetric unit membrane during urothelial differentiation.Mol Biol Rep. 1996;23(1):3-11. doi: 10.1007/BF00357068. Mol Biol Rep. 1996. PMID: 8983014 Review.
Cited by
-
Selective targeting of lectins and their macropinocytosis in urothelial tumours: translation from in vitro to ex vivo.Histochem Cell Biol. 2023 Nov;160(5):435-452. doi: 10.1007/s00418-023-02224-2. Epub 2023 Aug 3. Histochem Cell Biol. 2023. PMID: 37535087 Free PMC article.
-
Freeze-fracture replica immunolabelling reveals urothelial plaques in cultured urothelial cells.PLoS One. 2012;7(6):e38509. doi: 10.1371/journal.pone.0038509. Epub 2012 Jun 29. PLoS One. 2012. PMID: 22768045 Free PMC article.
-
Immunohistochemistry as a paramount tool in research of normal urothelium, bladder cancer and bladder pain syndrome.Eur J Histochem. 2021 Mar 24;65(2):3242. doi: 10.4081/ejh.2021.3242. Eur J Histochem. 2021. PMID: 33764020 Free PMC article. Review.
-
MAL facilitates the incorporation of exocytic uroplakin-delivering vesicles into the apical membrane of urothelial umbrella cells.Mol Biol Cell. 2012 Apr;23(7):1354-66. doi: 10.1091/mbc.E11-09-0823. Epub 2012 Feb 9. Mol Biol Cell. 2012. PMID: 22323295 Free PMC article.
-
Heterogeneity of uroplakin localization in human normal urothelium, papilloma and papillary carcinoma.Radiol Oncol. 2013 Oct 8;47(4):338-45. doi: 10.2478/raon-2013-0052. eCollection 2013. Radiol Oncol. 2013. PMID: 24294178 Free PMC article.
References
-
- Hicks RM. The mammalian urinary bladder: an accommodating organ. Biol Rev Camb Philos Soc. 1975;50:215–246. - PubMed
-
- Romih R, Korosec P, de Mello W, Jr, Jezernik K. Differentiation of epithelial cells in the urinary tract. Cell Tissue Res. 2005;320:259–268. - PubMed
-
- Veranic P, Romih R, Jezernik K. What determines differentiation of urothelial umbrella cells? Eur J Cell Biol. 2004;83:27–34. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

