Functional CD8 T cell memory responding to persistent latent infection is maintained for life
- PMID: 21890658
- PMCID: PMC4102748
- DOI: 10.4049/jimmunol.1100666
Functional CD8 T cell memory responding to persistent latent infection is maintained for life
Abstract
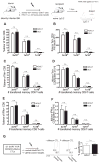
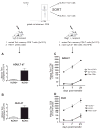
Aging is associated with depressed naive T cell responses, but it is less clear whether T cell memory established early in life also becomes impaired with age. This is particularly important for T cells responding to latent persistent infection, which need to remain functional and capable of controlling the infection over the lifetime; however, repeated stimulation over the lifetime may dysregulate their maintenance or function, potentially contributing to impaired immunity in the elderly. Systemic infection with HSV-1, a persistent latent virus, is associated with memory inflation of virus-specific CD8 T cells. We tested how these inflated memory cells are maintained from adulthood into old age. We found no significant differences in the numbers (i.e., blood, spleen), ex vivo Ag-specific IFN-γ production, and in vivo recall response to HSV-1 (i.e., proliferation, IFN-γ production, cytolysis) between adult and old memory T cells. There was a discrete shift from dominantly effector memory phenotype in the adults to a central memory-like phenotype in the old mice, with fewer old cells expressing the killer cell lectin-like receptor G1 (KLRG1). Adult and old KLRG1(+) memory CD8 T cells were functionally identical: both produced IFN-γ but could minimally proliferate in response to viral challenge. Interestingly, regardless of age, KLRG1(+) cells retained the ability to proliferate and survive in response to homeostatic signals, both in vitro (culture with IL-7 and IL-15) and in vivo (expansion following transfer into lymphopenic recipients). This finding demonstrates that functional effector memory T cells, including those expressing KLRG-1, are maintained and are functional for life, despite the presence of persistent viral infection.
Figures

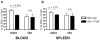



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

