The inflammasome adaptor ASC regulates the function of adaptive immune cells by controlling Dock2-mediated Rac activation and actin polymerization
- PMID: 21892172
- PMCID: PMC3178750
- DOI: 10.1038/ni.2095
The inflammasome adaptor ASC regulates the function of adaptive immune cells by controlling Dock2-mediated Rac activation and actin polymerization
Abstract
The adaptor ASC contributes to innate immunity through the assembly of inflammasome complexes that activate the cysteine protease caspase-1. Here we demonstrate that ASC has an inflammasome-independent, cell-intrinsic role in cells of the adaptive immune response. ASC-deficient mice showed defective antigen presentation by dendritic cells (DCs) and lymphocyte migration due to impaired actin polymerization mediated by the small GTPase Rac. Genome-wide analysis showed that ASC, but not the cytoplasmic receptor NLRP3 or caspase-1, controlled the mRNA stability and expression of Dock2, a guanine nucleotide-exchange factor that mediates Rac-dependent signaling in cells of the immune response. Dock2-deficient DCs showed defective antigen uptake similar to that of ASC-deficient cells. Ectopic expression of Dock2 in ASC-deficient cells restored Rac-mediated actin polymerization, antigen uptake and chemotaxis. Thus, ASC shapes adaptive immunity independently of inflammasomes by modulating Dock2-dependent Rac activation and actin polymerization in DCs and lymphocytes.
Figures
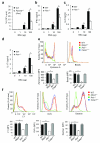
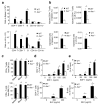
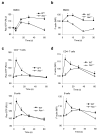
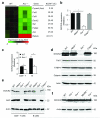
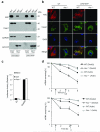
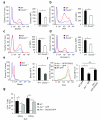
Comment in
-
ASCertaining cytoskeletal rearrangements in antigen presentation and migration.Nat Immunol. 2011 Sep 20;12(10):923-5. doi: 10.1038/ni.2114. Nat Immunol. 2011. PMID: 21934669
References
-
- Bauer C, et al. Colitis induced in mice with dextran sulfate sodium (DSS) is mediated by the NLRP3 inflammasome. Gut. 2010;59:1192–1199. - PubMed
-
- Dupaul-Chicoine J, et al. Control of intestinal homeostasis, colitis, and colitis-associated colorectal cancer by the inflammatory caspases. Immunity. 2010;32:367–378. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

