Multiple Myeloma Includes Phenotypically Defined Subsets of Clonotypic CD20+ B Cells that Persist During Treatment with Rituximab
- PMID: 21892289
- PMCID: PMC3161648
- DOI: 10.4137/cmo.s615
Multiple Myeloma Includes Phenotypically Defined Subsets of Clonotypic CD20+ B Cells that Persist During Treatment with Rituximab
Abstract
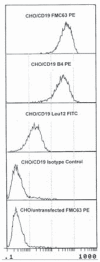
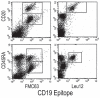
Potential progenitor B cell compartments in multiple myeloma (MM) are clinically important. MM B cells and some circulating MM plasma cells express CD20, predicting their clearance by treatment with anti-CD20. Here we describe two types of clonotypic CD20+ B cell in peripheral blood of myeloma patients, identified by their expression of CD19 and CD20 epitopes, their expression of CD45RA and their light scatter properties. Thus, the circulating component of the MM clone includes at least two distinct CD19+ CD20+ B cell compartments, as well as CD138+ CD20+ plasma cells. To determine whether either or both B cell subsets and the CD20+ plasma cell subset were depleted by anti-CD20 therapy, they were evaluated before, during and after treatment of patients with rituximab (anti-CD20), followed by quantifying B cell subsets over a 5 month period during and after treatment. Overall, all three types of circulating B lineage cells persist despite treatment with rituximab. The inability of rituximab to prolong survival in MM may result from this failure to deplete CD20+ B and plasma cells in MM.
Keywords: B cell depletion by rituximab; B lineage cells; CD19 epitopes; CD20 epitopes; multiple myeloma.
Figures





References
-
- Bakkus MH, Van Riet C, Van Camp I, Thielemann B. Evidence that the clonogenic cell in multiple myeloma originates from a pre-switched but somatically mutated B. cell. Brit J Hematol. 87:68–74. - PubMed
-
- Bergsagel PL, Masellis S, Szczepek A, Mant MJ, Belch AR, Pilarski LM. In multiple myeloma, clonotypic B. lymphocytes are detectable among CD19+ peripheral blood cells expressing CD38, CD56 and monotypic immunoglobulin light chain. Blood. 1995;85:436–47. - PubMed
-
- Bonavida B. Rituximab-induced inhibition of antiapoptotic cell survival pathways: implications in chemo/immunoresistance, rituximab unresponsiveness, prognostic and novel therapeutic interventions. Oncogene. 2007;26(25):3629–36. - PubMed
-
- Byrd JC, Kitada S, Flinn IW, Aron JL, Pearson M, Lucas D, Reed JC. The mechanism of tumor cell clearance by rituximab in vivo in patients with B.-cell chronic lymphocytic leukemia: evidence of caspase activation and apoptosis induction. Blood. 2002;99(3):1038–43. - PubMed
LinkOut - more resources
Full Text Sources

