Starch-binding domain-containing protein 1 (Stbd1) and glycogen metabolism: Identification of the Atg8 family interacting motif (AIM) in Stbd1 required for interaction with GABARAPL1
- PMID: 21893048
- PMCID: PMC3411280
- DOI: 10.1016/j.bbrc.2011.08.106
Starch-binding domain-containing protein 1 (Stbd1) and glycogen metabolism: Identification of the Atg8 family interacting motif (AIM) in Stbd1 required for interaction with GABARAPL1
Abstract
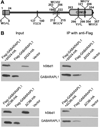
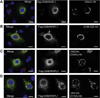
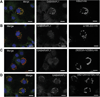
Glycogen, a branched polymer of glucose, acts as an intracellular carbon and energy reserve in many tissues and cell types. An important pathway for its degradation is by transport to lysosomes in an autophagy-like process. It has been proposed that starch-binding domain-containing protein 1 (Stbd1) may participate in this mechanism by anchoring glycogen to intracellular membranes. In addition, Stbd1 has been reported to interact with a known autophagy protein, GABARAPL1, a member of the Atg8 family. Here, we confirm this interaction and identify an Atg8 interacting motif (AIM) in Stbd1 necessary for GABARAPL1 binding as judged by co-immunoprecipitation from cell extracts and co-localization in cells as evidenced by immunofluorescence microscopy. The AIM sequence of Stbd1 (200)HEEWEMV(206) lies within a predicted disordered region of the molecule and fits the consensus of other AIM sequences in cargo-specifying proteins such as p62 and Nix. Mutation of the AIM, including single point mutations of either W203 or V206, eliminated the co-localization of Stbd1 with both over-expressed and endogenous GABARAPL1. Stbd1 may therefore function as a novel cargo binding protein that delivers glycogen to lysosomes in an autophagic pathway that could be termed "glycophagy".
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures

References
-
- Roach PJ. Glycogen and its metabolism. Curr. Mol. Med. 2002;2:101–120. - PubMed
-
- Greenberg CC, Jurczak MJ, Danos AM, Brady MJ. Glycogen branches out: new perspectives on the role of glycogen metabolism in the integration of metabolic pathways. Am. J. Physiol. Endocrinol. Metab. 2006;291:E1–E8. - PubMed
-
- Shearer J, Graham TE. Novel aspects of skeletal muscle glycogen and its regulation during rest and exercise. Exerc. Sport Sci. Rev. 2004;32:120–126. - PubMed
-
- Rosenfeld EL. Alpha-glucosidases (gamma-amylases) in human and animal organisms. Pathol. Biol. (Paris) 1975;23:71–84. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

