Synthetic double-stranded RNA enhances airway inflammation and remodelling in a rat model of asthma
- PMID: 21896009
- PMCID: PMC3194222
- DOI: 10.1111/j.1365-2567.2011.03473.x
Synthetic double-stranded RNA enhances airway inflammation and remodelling in a rat model of asthma
Abstract
Respiratory viral infections are frequently associated with exacerbations of asthma. Double-stranded RNA (dsRNA) produced during viral infections may be one of the stimuli for exacerbation. We aimed to assess the potential effect of dsRNA on certain aspects of chronic asthma through the administration of polyinosine-polycytidylic acid (poly I:C), synthetic dsRNA, to a rat model of asthma. Brown Norway rats were sensitized to ovalbumin and challenged three times to evoke airway remodelling. The effect of poly I:C on the ovalbumin-induced airway inflammation and structural changes was assessed from bronchoalveolar lavage fluid and histological findings. The expression of cytokines and chemokines was evaluated by real-time quantitative reverse transcription PCR and ELISA. Ovalbumin-challenged animals showed an increased number of total cells and eosinophils in bronchoalveolar lavage fluid compared with PBS-challenged controls. Ovalbumin-challenged animals treated with poly I:C showed an increased number of total cells and neutrophils in bronchoalveolar lavage fluid compared with those without poly I:C treatment. Ovalbumin-challenged animals showed goblet cell hyperplasia, increased airway smooth muscle mass, and proliferation of both airway epithelial cells and airway smooth muscle cells. Treatment with poly I:C enhanced these structural changes. Among the cytokines and chemokines examined, the expression of interleukins 12 and 17 and of transforming growth factor-β(1) in ovalbumin-challenged animals treated with poly I:C was significantly increased compared with those of the other groups. Double-stranded RNA enhanced airway inflammation and remodelling in a rat model of bronchial asthma. These observations suggest that viral infections may promote airway remodelling.
© 2011 The Authors. Immunology © 2011 Blackwell Publishing Ltd.
Figures

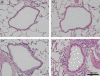
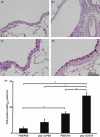
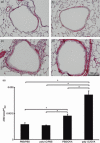
 ) (e). Data are presented as means (SEM). *P < 0·05.
) (e). Data are presented as means (SEM). *P < 0·05.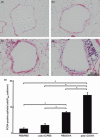
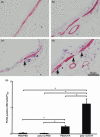
 ) (e). Data are presented as means (SEM). *P < 0·05.
) (e). Data are presented as means (SEM). *P < 0·05.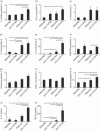
Similar articles
-
The presence of LPS in OVA inhalations affects airway inflammation and AHR but not remodeling in a rodent model of asthma.Am J Physiol Lung Cell Mol Physiol. 2012 Jul 1;303(1):L54-63. doi: 10.1152/ajplung.00208.2011. Epub 2012 Apr 20. Am J Physiol Lung Cell Mol Physiol. 2012. PMID: 22523281
-
Effect of TLR3/dsRNA complex inhibitor on Poly(I:C)-induced airway inflammation in Swiss albino mice.Environ Sci Pollut Res Int. 2023 Feb;30(10):28118-28132. doi: 10.1007/s11356-022-23987-6. Epub 2022 Nov 17. Environ Sci Pollut Res Int. 2023. PMID: 36394807
-
Sites of allergic airway smooth muscle remodeling and hyperresponsiveness are not associated in the rat.J Appl Physiol (1985). 2010 Oct;109(4):1170-8. doi: 10.1152/japplphysiol.01168.2009. Epub 2010 Jul 22. J Appl Physiol (1985). 2010. PMID: 20651225
-
Aspergillus fumigatus-Host Interactions Mediating Airway Wall Remodelling in Asthma.J Fungi (Basel). 2022 Feb 6;8(2):159. doi: 10.3390/jof8020159. J Fungi (Basel). 2022. PMID: 35205913 Free PMC article. Review.
-
The role of neutrophils in chronic cough.Hum Cell. 2024 Sep;37(5):1316-1324. doi: 10.1007/s13577-024-01089-4. Epub 2024 Jun 24. Hum Cell. 2024. PMID: 38913146 Review.
Cited by
-
Mouse Model of IL-17-Dominant Rhinitis Using Polyinosinic-Polycytidylic Acid.Allergy Asthma Immunol Res. 2017 Nov;9(6):540-549. doi: 10.4168/aair.2017.9.6.540. Allergy Asthma Immunol Res. 2017. PMID: 28913994 Free PMC article.
-
Mendelian randomization analysis reveals causal relationship between allergic diseases and influenza.World Allergy Organ J. 2025 Jun 7;18(7):101077. doi: 10.1016/j.waojou.2025.101077. eCollection 2025 Jul. World Allergy Organ J. 2025. PMID: 40529241 Free PMC article.
-
Poly I:C Exacerbates Airway Inflammation and Remodeling in Cigarette Smoke-Exposed Mice.Lung. 2022 Dec;200(6):677-686. doi: 10.1007/s00408-022-00574-7. Epub 2022 Oct 21. Lung. 2022. PMID: 36269393 Free PMC article.
-
Effect of low-level laser therapy on allergic asthma in rats.Lasers Med Sci. 2014 May;29(3):1043-50. doi: 10.1007/s10103-013-1456-5. Epub 2013 Oct 26. Lasers Med Sci. 2014. PMID: 24158722
-
Influenza in Asthmatics: For Better or for Worse?Front Immunol. 2018 Aug 10;9:1843. doi: 10.3389/fimmu.2018.01843. eCollection 2018. Front Immunol. 2018. PMID: 30147697 Free PMC article. Review.
References
-
- Bousquet J, Jeffery PK, Busse WW, Johnson M, Vignola AM. Asthma. From bronchoconstriction to airways inflammation and remodeling. Am J Respir Crit Care Med. 2000;161:1720–45. - PubMed
-
- Atmar RL, Guy E, Guntupalli KK, Zimmerman JL, Bandi VD, Baxter BD, Greenberg SB. Respiratory tract viral infections in inner-city asthmatic adults. Arch Intern Med. 1998;158:2453–9. - PubMed
-
- Grissell TV, Powell H, Shafren DR, Boyle MJ, Hensley MJ, Jones PD, Whitehead BF, Gibson PG. Interleukin-10 gene expression in acute virus-induced asthma. Am J Respir Crit Care Med. 2005;172:433–9. - PubMed
-
- Alexopoulou L, Holt AC, Medzhitov R, Flavell RA. Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature. 2001;413:732–8. - PubMed

