The interplay between transient α-helix formation and side chain rotamer distributions in disordered proteins probed by methyl chemical shifts
- PMID: 21898648
- PMCID: PMC3302646
- DOI: 10.1002/pro.726
The interplay between transient α-helix formation and side chain rotamer distributions in disordered proteins probed by methyl chemical shifts
Abstract
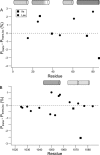
The peptide backbones of disordered proteins are routinely characterized by NMR with respect to transient structure and dynamics. Little experimental information is, however, available about the side chain conformations and how structure in the backbone affects the side chains. Methyl chemical shifts can in principle report the conformations of aliphatic side chains in disordered proteins and in order to examine this two model systems were chosen: the acid denatured state of acyl-CoA binding protein (ACBP) and the intrinsically disordered activation domain of the activator for thyroid hormone and retinoid receptors (ACTR). We find that small differences in the methyl carbon chemical shifts due to the γ-gauche effect may provide information about the side chain rotamer distributions. However, the effects of neighboring residues on the methyl group chemical shifts obscure the direct observation of γ-gauche effect. To overcome this, we reference the chemical shifts to those in a more disordered state resulting in residue specific random coil chemical shifts. The (13)C secondary chemical shifts of the methyl groups of valine, leucine, and isoleucine show sequence specific effects, which allow a quantitative analysis of the ensemble of χ(2)-angles of especially leucine residues in disordered proteins. The changes in the rotamer distributions upon denaturation correlate to the changes upon helix induction by the co-solvent trifluoroethanol, suggesting that the side chain conformers are directly or indirectly related to formation of transient α-helices.
Copyright © 2011 The Protein Society.
Figures
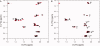


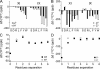
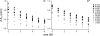

Similar articles
-
Accurate measurement of methyl 13C chemical shifts by solid-state NMR for the determination of protein side chain conformation: the influenza a M2 transmembrane peptide as an example.J Am Chem Soc. 2009 Jun 10;131(22):7806-16. doi: 10.1021/ja901550q. J Am Chem Soc. 2009. PMID: 19441789 Free PMC article.
-
Isoleucine Side Chains as Reporters of Conformational Freedom in Protein Folding Studied by DNP-Enhanced NMR.J Am Chem Soc. 2025 May 7;147(18):15867-15879. doi: 10.1021/jacs.5c04159. Epub 2025 Apr 26. J Am Chem Soc. 2025. PMID: 40285725 Free PMC article.
-
Determining valine side-chain rotamer conformations in proteins from methyl 13C chemical shifts: application to the 360 kDa half-proteasome.J Am Chem Soc. 2011 Jun 1;133(21):8272-81. doi: 10.1021/ja2014532. Epub 2011 May 5. J Am Chem Soc. 2011. PMID: 21545099
-
Helical propensity in an intrinsically disordered protein accelerates ligand binding.Angew Chem Int Ed Engl. 2014 Feb 3;53(6):1548-51. doi: 10.1002/anie.201307712. Epub 2014 Jan 21. Angew Chem Int Ed Engl. 2014. PMID: 24449148
-
Unraveling the meaning of chemical shifts in protein NMR.Biochim Biophys Acta Proteins Proteom. 2017 Nov;1865(11 Pt B):1564-1576. doi: 10.1016/j.bbapap.2017.07.005. Epub 2017 Jul 15. Biochim Biophys Acta Proteins Proteom. 2017. PMID: 28716441 Review.
Cited by
-
Pressure dependence of side chain 13C chemical shifts in model peptides Ac-Gly-Gly-Xxx-Ala-NH2.J Biomol NMR. 2017 Oct;69(2):53-67. doi: 10.1007/s10858-017-0134-5. Epub 2017 Sep 14. J Biomol NMR. 2017. PMID: 28913741
-
Fusidic acid resistance through changes in the dynamics of the drug target.Proc Natl Acad Sci U S A. 2020 Oct 13;117(41):25523-25531. doi: 10.1073/pnas.2008577117. Epub 2020 Sep 30. Proc Natl Acad Sci U S A. 2020. PMID: 32999060 Free PMC article.
-
Is a malleable protein necessarily highly dynamic? The hydrophobic core of the nuclear coactivator binding domain is well ordered.Biophys J. 2012 Apr 4;102(7):1627-35. doi: 10.1016/j.bpj.2012.02.014. Epub 2012 Apr 3. Biophys J. 2012. PMID: 22500763 Free PMC article.
-
Ab initio calculations of possible γ-gauche effects in the ¹³C-NMR for methine and carbonyl carbons in precise polyethylene acrylic acid copolymers.Molecules. 2013 Jul 29;18(8):9010-20. doi: 10.3390/molecules18089010. Molecules. 2013. PMID: 23899834 Free PMC article.
References
-
- Dyson HJ, Wright PE. Intrinsically unstructured proteins and their functions. Nat Rev Mol Cell Bio. 2005;6:197–208. - PubMed
-
- Shortle D. The denatured state (the other half of the folding equation) and its role in protein stability. FASEB J. 1996;10:27–34. - PubMed
-
- Jensen MR, Salmon L, Nodet G, Blackledge M. Defining conformational ensembles of intrinsically disordered and partially folded proteins directly from chemical shifts. J Am Chem Soc. 2010;132:1270–1272. - PubMed
-
- Gillespie JR, Shortle D. Characterization of long-range structure in the denatured state of staphylococcal nuclease. I. Paramagnetic relaxation enhancement by nitroxide spin labels. J Mol Biol. 1997;268:158–169. - PubMed
-
- Lietzow MA, Jamin M, Dyson HJ, Wright PE. Mapping long-range contacts in a highly unfolded protein. J Mol Biol. 2002;322:655–662. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

