A Ca(2+) threshold for induction of spike-timing-dependent depression in the mouse striatum
- PMID: 21900580
- PMCID: PMC6623411
- DOI: 10.1523/JNEUROSCI.3206-11.2011
A Ca(2+) threshold for induction of spike-timing-dependent depression in the mouse striatum
Abstract
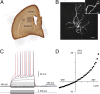
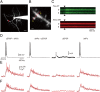
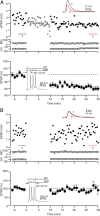
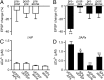
The striatum is the principal input nucleus of the basal ganglia, receiving glutamatergic afferents from the cerebral cortex. There is much interest in mechanisms of synaptic plasticity in the corticostriatal synapses. We used two-photon microscopy and whole-cell recording to measure changes in intracellular calcium concentration ([Ca(2+)](i)) associated with spike-time-dependent plasticity in mouse striatum. Uncaging glutamate adjacent to a dendritic spine caused a postsynaptic potential at the soma and a rise in spine [Ca(2+)](i). Action potentials elicited at the soma raised both dendrite and spine [Ca(2+)](i). Pairing protocols in which glutamate uncaging preceded action potentials by 10 ms (pre-post protocol) produced supralinear increases in spine [Ca(2+)](i) compared with the sum of increases seen with uncaging and action potentials alone, or timing protocols in which the uncaging followed the action potentials (post-pre protocols). The supralinear component of the increases in [Ca(2+)](i) were eliminated by the voltage-sensitive calcium channel blocker nimodipine. In the adjacent parent dendrites, the increases in [Ca(2+)](i) were neither supralinear nor sensitive to the relative pre-post timing. In parallel experiments, we investigated the effects of these pairing protocols on spike-timing-dependent synaptic plasticity. Long-term depression (t-LTD) of corticostriatal inputs was induced by pre-post but not post-pre protocols. Intracellular calcium chelators and calcium antagonists blocked pre-post t-LTD, confirming that elevated calcium entering via voltage-sensitive calcium channels is necessary for t-LTD. These findings confirm a spine [Ca(2+)](i) threshold for induction of t-LTD in the corticostriatal pathway, mediated by the supralinear increase in [Ca(2+)](i) associated with pre-post induction protocols.
Figures



References
-
- Bauswein E, Fromm C, Preuss A. Corticostriatal cells in comparison with pyramidal tract neurons: contrasting properties in the behaving monkey. Brain Res. 1989;493:198–203. - PubMed
-
- Bonsi P, Pisani A, Bernardi G, Calabresi P. Stimulus frequency, calcium levels and striatal synaptic plasticity. Neuroreport. 2003;14:419–422. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous
