Rapid plasticity of visual responses in the adult lateral geniculate nucleus
- PMID: 21903075
- PMCID: PMC3170518
- DOI: 10.1016/j.neuron.2011.06.025
Rapid plasticity of visual responses in the adult lateral geniculate nucleus
Abstract
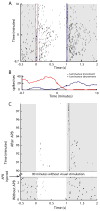
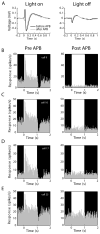
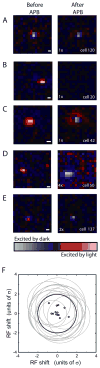
Compared to the developing visual system, where neuronal plasticity has been well characterized at multiple levels, little is known about plasticity in the adult, particularly within subcortical structures. We made intraocular injections of 2-amino-4-phosphonobutyric acid (APB) in adult cats to block visual responses in On-center retinal ganglion cells and examined the consequences on visual responses in the lateral geniculate nucleus (LGN) of the thalamus. In contrast to current views of retinogeniculate organization, which hold that On-center LGN neurons should become silent with APB, we find that ∼50% of On-center neurons rapidly develop Off-center responses. The time course of these emergent responses and the actions of APB in the retina indicate the plasticity occurs within the LGN. These results suggest there is greater divergence of retinogeniculate connections than previously recognized and that functionally silent, nonspecific retinal inputs can serve as a substrate for rapid plasticity in the adult.
Copyright © 2011 Elsevier Inc. All rights reserved.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

