The regulated secretory pathway in CD4(+) T cells contributes to human immunodeficiency virus type-1 cell-to-cell spread at the virological synapse
- PMID: 21909273
- PMCID: PMC3164651
- DOI: 10.1371/journal.ppat.1002226
The regulated secretory pathway in CD4(+) T cells contributes to human immunodeficiency virus type-1 cell-to-cell spread at the virological synapse
Abstract
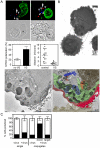
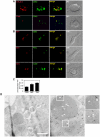
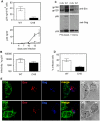
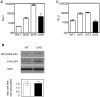
Direct cell-cell spread of Human Immunodeficiency Virus type-1 (HIV-1) at the virological synapse (VS) is an efficient mode of dissemination between CD4(+) T cells but the mechanisms by which HIV-1 proteins are directed towards intercellular contacts is unclear. We have used confocal microscopy and electron tomography coupled with functional virology and cell biology of primary CD4(+) T cells from normal individuals and patients with Chediak-Higashi Syndrome and report that the HIV-1 VS displays a regulated secretion phenotype that shares features with polarized secretion at the T cell immunological synapse (IS). Cell-cell contact at the VS re-orientates the microtubule organizing center (MTOC) and organelles within the HIV-1-infected T cell towards the engaged target T cell, concomitant with polarization of viral proteins. Directed secretion of proteins at the T cell IS requires specialized organelles termed secretory lysosomes (SL) and we show that the HIV-1 envelope glycoprotein (Env) localizes with CTLA-4 and FasL in SL-related compartments and at the VS. Finally, CD4(+) T cells that are disabled for regulated secretion are less able to support productive cell-to-cell HIV-1 spread. We propose that HIV-1 hijacks the regulated secretory pathway of CD4(+) T cells to enhance its dissemination.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




Similar articles
-
LFA-1 Engagement Triggers T Cell Polarization at the HIV-1 Virological Synapse.J Virol. 2016 Oct 14;90(21):9841-9854. doi: 10.1128/JVI.01152-16. Print 2016 Nov 1. J Virol. 2016. PMID: 27558417 Free PMC article.
-
The infectious synapse formed between mature dendritic cells and CD4(+) T cells is independent of the presence of the HIV-1 envelope glycoprotein.Retrovirology. 2013 Apr 16;10:42. doi: 10.1186/1742-4690-10-42. Retrovirology. 2013. PMID: 23590845 Free PMC article.
-
Predominant mode of human immunodeficiency virus transfer between T cells is mediated by sustained Env-dependent neutralization-resistant virological synapses.J Virol. 2007 Nov;81(22):12582-95. doi: 10.1128/JVI.00381-07. Epub 2007 Aug 29. J Virol. 2007. PMID: 17728240 Free PMC article.
-
Cell-to-Cell Spread of HIV and Viral Pathogenesis.Adv Virus Res. 2016;95:43-85. doi: 10.1016/bs.aivir.2016.03.001. Epub 2016 Apr 4. Adv Virus Res. 2016. PMID: 27112280 Review.
-
Mechanisms of enhanced HIV spread through T-cell virological synapses.Immunol Rev. 2013 Jan;251(1):113-24. doi: 10.1111/imr.12022. Immunol Rev. 2013. PMID: 23278744 Review.
Cited by
-
Pathways of cell-cell transmission of HTLV-1.Front Microbiol. 2012 Oct 24;3:378. doi: 10.3389/fmicb.2012.00378. eCollection 2012. Front Microbiol. 2012. PMID: 23109932 Free PMC article.
-
Equine Herpesvirus 1 Bridles T Lymphocytes To Reach Its Target Organs.J Virol. 2019 Mar 21;93(7):e02098-18. doi: 10.1128/JVI.02098-18. Print 2019 Apr 1. J Virol. 2019. PMID: 30651370 Free PMC article.
-
Murine leukemia virus Gag localizes to the uropod of migrating primary lymphocytes.J Virol. 2014 Sep;88(18):10541-55. doi: 10.1128/JVI.01104-14. Epub 2014 Jun 25. J Virol. 2014. PMID: 24965475 Free PMC article.
-
Which Antibody Functions are Important for an HIV Vaccine?Front Immunol. 2014 Jun 18;5:289. doi: 10.3389/fimmu.2014.00289. eCollection 2014. Front Immunol. 2014. PMID: 24995008 Free PMC article. Review.
-
HIV cell-to-cell transmission requires the production of infectious virus particles and does not proceed through env-mediated fusion pores.J Virol. 2012 Apr;86(7):3924-33. doi: 10.1128/JVI.06478-11. Epub 2012 Jan 18. J Virol. 2012. PMID: 22258237 Free PMC article.
References
-
- Sattentau Q. Avoiding the void: cell-to-cell spread of human viruses. Nat Rev Microbiol. 2008;6:815–826. - PubMed
-
- Sowinski S, Jolly C, Berninghausen O, Purbhoo MA, Chauveau A, et al. Membrane nanotubes physically connect T cells over long distances presenting a novel route for HIV-1 transmission. Nat Cell Biol. 2008;10:211–219. - PubMed
-
- Jolly C, Sattentau QJ. Retroviral spread by induction of virological synapses. Traffic. 2004;5:643–650. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

