The evolutionarily conserved longevity determinants HCF-1 and SIR-2.1/SIRT1 collaborate to regulate DAF-16/FOXO
- PMID: 21909281
- PMCID: PMC3164695
- DOI: 10.1371/journal.pgen.1002235
The evolutionarily conserved longevity determinants HCF-1 and SIR-2.1/SIRT1 collaborate to regulate DAF-16/FOXO
Abstract
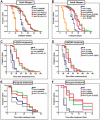
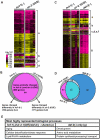
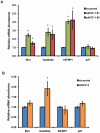
The conserved DAF-16/FOXO transcription factors and SIR-2.1/SIRT1 deacetylases are critical for diverse biological processes, particularly longevity and stress response; and complex regulation of DAF-16/FOXO by SIR-2.1/SIRT1 is central to appropriate biological outcomes. Caenorhabditis elegans Host Cell Factor 1 (HCF-1) is a longevity determinant previously shown to act as a co-repressor of DAF-16. We report here that HCF-1 represents an integral player in the regulatory loop linking SIR-2.1/SIRT1 and DAF-16/FOXO in both worms and mammals. Genetic analyses showed that hcf-1 acts downstream of sir-2.1 to influence lifespan and oxidative stress response in C. elegans. Gene expression profiling revealed a striking 80% overlap between the DAF-16 target genes responsive to hcf-1 mutation and sir-2.1 overexpression. Subsequent GO-term analyses of HCF-1 and SIR-2.1-coregulated DAF-16 targets suggested that HCF-1 and SIR-2.1 together regulate specific aspects of DAF-16-mediated transcription particularly important for aging and stress responses. Analogous to its role in regulating DAF-16/SIR-2.1 target genes in C. elegans, the mammalian HCF-1 also repressed the expression of several FOXO/SIRT1 target genes. Protein-protein association studies demonstrated that SIR-2.1/SIRT1 and HCF-1 form protein complexes in worms and mammalian cells, highlighting the conservation of their regulatory relationship. Our findings uncover a conserved interaction between the key longevity determinants SIR-2.1/SIRT1 and HCF-1, and they provide new insights into the complex regulation of FOXO proteins.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


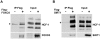
References
-
- Kenyon CJ. The genetics of ageing. Nature. 2010;464:504–512. - PubMed
-
- Burgering BM, Kops GJ. Cell cycle and death control: long live Forkheads. Trends Biochem Sci. 2002;27:352–360. - PubMed
-
- Lin K, Dorman JB, Rodan A, Kenyon C. daf-16: An HNF-3/forkhead family member that can function to double the life-span of Caenorhabditis elegans. Science. 1997;278:1319–1322. - PubMed
-
- Giannakou ME, Goss M, Junger MA, Hafen E, Leevers SJ, et al. Long-lived Drosophila with overexpressed dFOXO in adult fat body. Science. 2004;305:361. - PubMed
-
- Hwangbo DS, Gershman B, Tu MP, Palmer M, Tatar M. Drosophila dFOXO controls lifespan and regulates insulin signalling in brain and fat body. Nature. 2004;429:562–566. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

