Novel synthesis of pseudopeptides bearing a difluoromethyl group by Ugi reaction and desulfanylation
- PMID: 21915210
- PMCID: PMC3170163
- DOI: 10.3762/bjoc.7.123
Novel synthesis of pseudopeptides bearing a difluoromethyl group by Ugi reaction and desulfanylation
Abstract
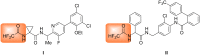
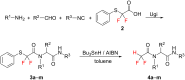
Thirteen difluoromethyl-containing pseudopeptides were synthesized by Ugi reaction using the novel building block 2,2-difluoro-2-(phenylthio)acetic acid (2) as one component, followed by removal of the phenylsulfanyl protecting group in the presence of tributyltin hydride and azobisisobutyronitrile.
Keywords: Ugi reaction; difluoromethyl functionality; gem-difluoromethylene-containing acid; pseudopeptides; reductive cleavage.
Figures

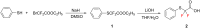
Similar articles
-
A general strategy for construction of a difluoromethyl compound library and its application in synthesis of pseudopeptides bearing a terminal difluoromethyl group.Org Biomol Chem. 2010 May 21;8(10):2386-91. doi: 10.1039/c000835d. Epub 2010 Mar 17. Org Biomol Chem. 2010. PMID: 20448896
-
A mild and efficient synthesis of monofluorinated α-lactam pseudopeptides via a novel dehydrofluorination of Ugi products.Mol Divers. 2010 Aug;14(3):501-6. doi: 10.1007/s11030-009-9209-x. Epub 2009 Nov 17. Mol Divers. 2010. PMID: 19921454
-
Difluorophenylsulfanylmethyl radical and difluoromethylene diradical synthons: gem-difluoromethylene building block.J Org Chem. 2004 Oct 1;69(20):6913-5. doi: 10.1021/jo0489768. J Org Chem. 2004. PMID: 15387624
-
Cyclic Imines in Ugi and Ugi-Type Reactions.ACS Comb Sci. 2020 Aug 10;22(8):361-400. doi: 10.1021/acscombsci.0c00046. Epub 2020 Jul 14. ACS Comb Sci. 2020. PMID: 32574488 Review.
-
Synthesis of N-heterocycles containing 1,5-disubstituted-1H-tetrazole via post-Ugi-azide reaction.Mol Divers. 2020 Aug;24(3):841-853. doi: 10.1007/s11030-019-09972-1. Epub 2019 Jun 20. Mol Divers. 2020. PMID: 31222498 Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
