Recent developments in gold-catalyzed cycloaddition reactions
- PMID: 21915211
- PMCID: PMC3167873
- DOI: 10.3762/bjoc.7.124
Recent developments in gold-catalyzed cycloaddition reactions
Abstract
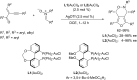
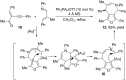
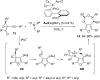
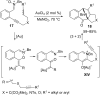
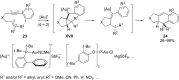
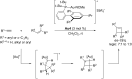
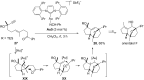
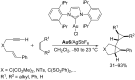
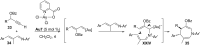
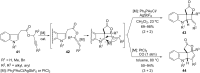
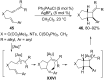
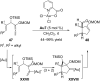
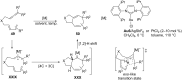
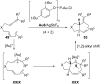
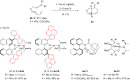
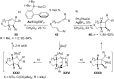
In the last years there have been extraordinary advances in the development of gold-catalyzed cycloaddition processes. In this review we will summarize some of the most remarkable examples, and present the mechanistic rational underlying the transformations.
Keywords: alkene; alkyne; allene; catalysis; cycloaddition; gold.
Figures

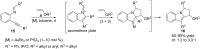
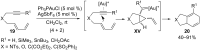
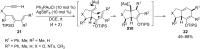
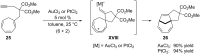
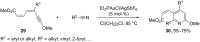


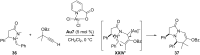
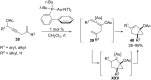
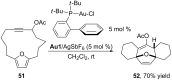
Similar articles
-
Rhodium-catalyzed acyloxy migration of propargylic esters in cycloadditions, inspiration from the recent "gold rush".Chem Soc Rev. 2012 Dec 7;41(23):7698-711. doi: 10.1039/c2cs35235d. Chem Soc Rev. 2012. PMID: 22895533 Free PMC article. Review.
-
Gold(I)-catalyzed enantioselective cycloaddition reactions.Beilstein J Org Chem. 2013 Oct 30;9:2250-64. doi: 10.3762/bjoc.9.264. Beilstein J Org Chem. 2013. PMID: 24204438 Free PMC article. Review.
-
Rhodium-Catalyzed [2+2+2] Cycloaddition Reactions of Linear Allene-Ene-Ynes to afford Fused Tricyclic Scaffolds: Insights into the Mechanism.Chemistry. 2017 Oct 20;23(59):14889-14899. doi: 10.1002/chem.201703194. Epub 2017 Sep 27. Chemistry. 2017. PMID: 28779482
-
Recent Advances in Gold(III) Chemistry: Structure, Bonding, Reactivity, and Role in Homogeneous Catalysis.Chem Rev. 2021 Jul 28;121(14):8364-8451. doi: 10.1021/acs.chemrev.0c00552. Epub 2020 Sep 23. Chem Rev. 2021. PMID: 32966741
-
Mechanistic understanding of [Rh(NHC)]-catalyzed intramolecular [5 + 2] cycloadditions of vinyloxiranes and vinylcyclopropanes with alkynes.Org Biomol Chem. 2018 Jun 13;16(23):4295-4303. doi: 10.1039/c8ob01117f. Org Biomol Chem. 2018. PMID: 29808214
Cited by
-
Enantioselective gold-catalyzed intermolecular [2+2] versus [4+2]-cycloadditions of 3-styrylindoles with N-allenamides: observation of interesting substituent effects.Chem Sci. 2015 Oct 1;6(10):5564-5570. doi: 10.1039/c5sc01827g. Epub 2015 Jun 23. Chem Sci. 2015. PMID: 29861893 Free PMC article.
-
Gold-catalyzed [4+3]- and [4+2]-annulations of 3-en-1-ynamides with isoxazoles via novel 6π-electrocyclizations of 3-azahepta trienyl cations.Chem Sci. 2018 Feb 19;9(11):2991-2995. doi: 10.1039/c8sc00232k. eCollection 2018 Mar 21. Chem Sci. 2018. PMID: 29732082 Free PMC article.
-
Gold-catalyzed cyclizations of cis-enediynes: insights into the nature of gold-aryne interactions.Angew Chem Int Ed Engl. 2013 Jul 22;52(30):7795-9. doi: 10.1002/anie.201301057. Epub 2013 Jun 20. Angew Chem Int Ed Engl. 2013. PMID: 23788447 Free PMC article.
-
Rhodium-catalyzed acyloxy migration of propargylic esters in cycloadditions, inspiration from the recent "gold rush".Chem Soc Rev. 2012 Dec 7;41(23):7698-711. doi: 10.1039/c2cs35235d. Chem Soc Rev. 2012. PMID: 22895533 Free PMC article. Review.
-
Origins of unique gold-catalysed chemo- and site-selective C-H functionalization of phenols with diazo compounds.Chem Sci. 2016 Mar 1;7(3):1988-1995. doi: 10.1039/c5sc04319k. Epub 2015 Nov 27. Chem Sci. 2016. PMID: 29899922 Free PMC article.
References
-
- Hudlicky T, Natchus M G. In: Organic Synthesis: Theory and Applications. Hudlicky T, editor. Vol. 2. Greenwich, CT: JAI; 1993. pp. 1–23.
-
- Carruthers W. Cycloaddition Reactions in Organic Synthesis. Oxford: Pergamon; 1990.
-
- Kobayashi S, Jorgensen K A. Cycloaddition Reactions in Organic Synthesis. Weinheim, Germany: Wiley-VCH; 2002.
LinkOut - more resources
Full Text Sources
