SIRT1 is essential for oncogenic signaling by estrogen/estrogen receptor α in breast cancer
- PMID: 21920899
- PMCID: PMC3206200
- DOI: 10.1158/0008-5472.CAN-11-1446
SIRT1 is essential for oncogenic signaling by estrogen/estrogen receptor α in breast cancer
Abstract
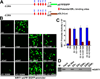
The NAD-dependent histone deacetylase silent information regulator 1 (SIRT1) is overexpressed and catalytically activated in a number of human cancers, but recent studies have actually suggested that it may function as a tumor suppressor and metastasis inhibitor in vivo. In breast cancer, SIRT1 stabilization has been suggested to contribute to the oncogenic potential of the estrogen receptor α (ERα), but SIRT1 activity has also been associated with ERα deacetylation and inactivation. In this study, we show that SIRT1 is critical for estrogen to promote breast cancer. ERα physically interacted and functionally cooperated with SIRT1 in breast cancer cells. ERα also bound to the promoter for SIRT1 and increased its transcription. SIRT1 expression induced by ERα was sufficient to activate antioxidant and prosurvival genes in breast cancer cells, such as catalase and glutathione peroxidase, and to inactivate tumor suppressor genes such as cyclin G2 (CCNG2) and p53. Moreover, SIRT1 inactivation eliminated estrogen/ERα-induced cell growth and tumor development, triggering apoptosis. Taken together, these results indicated that SIRT1 is required for estrogen-induced breast cancer growth. Our findings imply that the combination of SIRT1 inhibitors and antiestrogen compounds may offer more effective treatment strategies for breast cancer.
©2011 AACR.
Conflict of interest statement
All authors declare no conflict of interest.
Figures


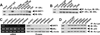



References
-
- Dali-Youcef N, Lagouge M, Froelich S, Koehl C, Schoonjans K, Auwerx J. Sirtuins: the ‘magnificient seven’, function, metabolism and longevity. Ann Med. 2007;39:335–345. - PubMed
-
- Haigis MC, Guarente LP. Mammalian sirtuins-emerging roles in physiology, aging, and calorie restriction. Genes Dev. 2006;20:2913–2921. - PubMed
-
- Huffman DM, Grizzle WE, Bamman MM, Kim JS, Eltoum IA, Elgavish A, Nagy TR. SIRT1 is significantly elevated in mouse and human prostate cancer. Cancer Res. 2007;67:6612–6618. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

