Drosophila, genetic screens, and cardiac function
- PMID: 21921272
- PMCID: PMC3678974
- DOI: 10.1161/CIRCRESAHA.111.244897
Drosophila, genetic screens, and cardiac function
Abstract
The fruit fly, Drosophila melanogaster, has been used to study genetics, development, and signaling for nearly a century, but only over the past few decades has this tremendous resource been the focus of cardiovascular research. Fly genetics offers sophisticated transgenic systems, molecularly defined genomic deficiencies, genome-wide transgenic RNAi lines, and numerous curated mutants to perform genetic screens. As a genetically tractable model, the fly facilitates gene discovery and can complement mammalian models of disease. The circulatory system in the fly comprises well-defined sets of cardiomyocytes, and methodological advances have permitted accurate characterization of cardiac morphology and function. Thus, fly genetics and genomics offer new approaches for gene discovery of adult cardiac phenotypes to identify evolutionarily conserved molecular signals that drive cardiovascular disease.
Figures

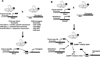

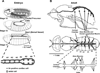

References
-
- Kohler RE. Lords of the fly : Drosophila genetics and the experimental life. Chicago: University of Chicago Press; 1994.
-
- Ashburner M, Golic KG, Hawley RS. Drosophila : A laboratory handbook. Cold Spring Harbor, N.Y.: Cold Spring Harbor Laboratory Press; 2005.
-
- Greenspan RJ. Fly pushing : The theory and practice of drosophila genetics. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory Press; 2004.
-
- Dahmann C. Drosophila : Methods and protocols. Totowa, N.J.: Humana Press; 2008.
-
- Brand AH, Perrimon N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development. 1993;118:401–415. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

