Using the Skindex-16 and Common Terminology Criteria for Adverse Events to assess rash symptoms: results of a pooled-analysis (N0993)
- PMID: 21922203
- PMCID: PMC3391325
- DOI: 10.1007/s00520-011-1266-x
Using the Skindex-16 and Common Terminology Criteria for Adverse Events to assess rash symptoms: results of a pooled-analysis (N0993)
Abstract
Background: Historically, skin toxicity has been assessed in prospective clinical trials using the clinician-reported National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE). The patient-reported Skindex-16 measures symptoms and perceptions of toxicity. This study was designed to compare information provided by these two measures.
Methods: Data were compiled from three placebo-controlled North Central Cancer Treatment Group studies (N06C4, N03CB, N05C4) having rash prevention as the primary objective. All used the Skindex-16 and CTCAE at baseline, weekly during treatment and during a minimum 2-week follow-up period. Statistical procedures, including Pearson correlations, were utilized to determine relationships between adverse event (AE) grades and Skindex-16 scores.
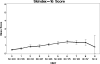
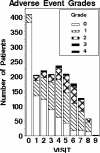
Results: Four hundred and twelve individual patients provided data (median age, 61; 134 male). Patients' Skindex-16 score results show a 0.9 overall mean (range 0-6 with 6 being worse symptoms), a 0.4 baseline mean (range, 0-4.3) and a 1.3 end-of-treatment mean (range, 0-5.9). Ninety-three, 142 and 177 patients experienced a grade 0, 1 and 2+ CTCAE skin toxicity, respectively. Baseline Skindex-16 scores had relatively low correlation with CTCAE grades. The correlation of rash grade with Skindex-16 scores ranged from r = 0.49 with the function subscale to r = 0.62 with the symptom subscale. The highest correlations of the maximum grade of any dermatological AE with the Skindex-16 were r = 0.48 for the total score and r = 0.55 for the symptom subscale.
Conclusions: The data reported support the decision to include both measures in a clinical trial to assess the patient experience, as each measure may specifically target varying symptoms and intensities.
Figures
References
-
- Hopewell JW. The skin: its structure and response to ionizing radiation. Int J of Radiation Biol. 1990;57:751–773. - PubMed
-
- Fu KK, Pajak TF, Trotti A, et al. A radiation therapy oncology group (RTOG) phase III randomized study to compare hyperfractionation and two variants of accelerated fractionation to standard fractionation radiotherapy for head and neck squamous cell carcinomas: first report of RTOG 9003. Int J Rad Oncol Biol Phys. 2000;48:7–16. - PubMed
-
- Archambeau JO, Pezner R, Wasserman T. Pathophysiology of irradiated skin and breast. Int J Rad Oncol Biol Phys. 1995;31:1171–1185. - PubMed
-
- Herbst RS, Maddox A-M, Rothenberg ML, et al. Selective oral epidermal growth factor receptor tyrosine kinase inhibitor ZD1839 is generally well tolerated and has activity in non-small cell lung cancer and other solid tumors: results of a phase I trial. J Clin Oncol. 2002;20:3815–3825. - PubMed
-
- Kris M, Natale RB, Herbst RS, et al. Efficacy of gefitinib, an inhibitor of the epidermal growth factor receptor tyrosine kinase, in symptomatic patients with non-small cell lung cancer. J Am Med Assoc. 2003;290:2149–2158. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical