DC mobilization from the skin requires docking to immobilized CCL21 on lymphatic endothelium and intralymphatic crawling
- PMID: 21930767
- PMCID: PMC3182054
- DOI: 10.1084/jem.20102392
DC mobilization from the skin requires docking to immobilized CCL21 on lymphatic endothelium and intralymphatic crawling
Abstract
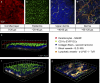
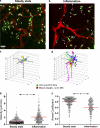
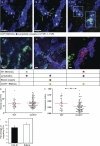
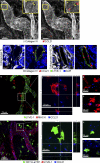
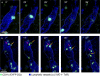
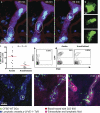
Dendritic cells (DCs) must travel through lymphatics to carry skin antigens into lymph nodes. The processes controlling their mobilization and migration have not been completely delineated. We studied how DCs in live mice respond to skin inflammation, transmigrate through lymphatic endothelium, and propagate in initial lymphatics. At steady state, dermal DCs remain sessile along blood vessels. Inflammation mobilizes them, accelerating their interstitial motility 2.5-fold. CCR7-deficient BMDCs crawl as fast as wild-type DCs but less persistently. We observed discrete depositions of CCL21 complexed with collagen-IV on the basement membrane of initial lymphatics. Activated DCs move directionally toward lymphatics, contact CCL21 puncta, and migrate through portals into the lumen. CCR7-deficient DCs arrive at lymphatics through random migration but fail to dock and transmigrate. Once inside vessels, wild-type DCs use lamellipodia to crawl along lymphatic endothelium and, sensing lymph flow, proceed downstream. DCs start drifting freely only in collecting lymphatics. These results demonstrate in vivo that the CCL21-CCR7 axis plays a dual role in DC mobilization: promoting both chemotaxis and arrest of DCs on lymphatic endothelium. Intralymphatic crawling, in which DCs combine active adhesion-based migration and directional cues from lymph flow, represents a new step in DC mobilization which may be amenable to regulation.
Figures