Vitiligo during Treatment of Crohn's Disease with Adalimumab: Adverse Effect or Co-Occurrence?
- PMID: 21931575
- PMCID: PMC3175360
- DOI: 10.1159/000324619
Vitiligo during Treatment of Crohn's Disease with Adalimumab: Adverse Effect or Co-Occurrence?
Abstract
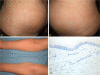
Adalimumab is a fully human monoclonal anti-tumor necrosis factor-a agent that is approved for the treatment of Crohn's disease. It has a good safety profile, injection site reactions being the most common adverse effect. We report a case of a 54-year-old woman with a 30-year history of Crohn's disease who developed achromic patches on the trunk and upper extremities after initiating treatment with adalimumab. Cutaneous biopsy confirmed diagnosis of vitiligo and laboratory testing ruled out thyroid disease. Concomitant occurrence of vitiligo and inflammatory bowel disease, although rare, has been described. A common autoimmune basis could explain this fact. Moreover, multiple cutaneous adverse effects have been described in the literature secondary to biologic treatments, including vitiligo. In this report, we discuss the possibility of vitiligo as secondary to adalimumab treatment or as an association between both diseases.
Keywords: Adalimumab; Anti-tumor necrosis factor-a; Cutaneous adverse effects; Vitiligo and Crohn's disease.
Figures
Similar articles
-
Management of inflammatory bowel disease with infliximab and other anti-tumor necrosis factor alpha therapies.BioDrugs. 2010 Dec 14;24 Suppl 1:3-14. doi: 10.2165/11586290-000000000-00000. BioDrugs. 2010. PMID: 21175228 Review.
-
Lymphomatoid Papulosis Type B in a Patient with Crohn's Disease Treated with TNF-Alpha Inhibitors Infliximab and Adalimumab.Acta Dermatovenerol Croat. 2019 Sep;27(3):202-204. Acta Dermatovenerol Croat. 2019. PMID: 31542070
-
Adalimumab - an effective and promising treatment for patients with fistulizing Crohn's disease: a case series.J Med Case Rep. 2011 Mar 19;5:109. doi: 10.1186/1752-1947-5-109. J Med Case Rep. 2011. PMID: 21418588 Free PMC article.
-
[Adalimumab for the treatment of adult Crohn's disease--update of a consensus report by the Working Group Inflammatory Bowel Disease of the Austrian Society of Gastroenterology and Hepatology].Z Gastroenterol. 2013 Sep;51(9):1101-9. doi: 10.1055/s-0033-1350165. Epub 2013 Aug 30. Z Gastroenterol. 2013. PMID: 23996653 German.
-
[Adalimumab for the treatment of Crohn's disease - consensus paper of the Working Group "chronic inflammatory bowel diseases" of the Austrian Society for Gastroenterology and Hepatology].Z Gastroenterol. 2009 Apr;47(4):372-80. doi: 10.1055/s-0028-1109218. Epub 2009 Apr 8. Z Gastroenterol. 2009. PMID: 19358065 German.
Cited by
-
Role of Cytokines in Vitiligo: Pathogenesis and Possible Targets for Old and New Treatments.Int J Mol Sci. 2021 Oct 22;22(21):11429. doi: 10.3390/ijms222111429. Int J Mol Sci. 2021. PMID: 34768860 Free PMC article. Review.
-
Development of Vitiligo during Treatment with Adalimumab: A Plausible or Paradoxical Response?Ann Dermatol. 2015 Oct;27(5):620-1. doi: 10.5021/ad.2015.27.5.620. Epub 2015 Oct 2. Ann Dermatol. 2015. PMID: 26512182 Free PMC article. No abstract available.
-
Vitiligo aggravated after major surgery for Crohn's disease: a consequence of severe surgical stress?Ann Gastroenterol. 2013;26(3):278-279. Ann Gastroenterol. 2013. PMID: 24714291 Free PMC article. No abstract available.
-
Seborrhoeic dermatitis and a herpes zoster infection developed during treatment with adalimumab due to Crohn's disease.Postepy Dermatol Alergol. 2015 Aug;32(4):317-9. doi: 10.5114/pdia.2014.44018. Epub 2015 Aug 12. Postepy Dermatol Alergol. 2015. PMID: 26366160 Free PMC article. No abstract available.
-
Vitiligo in a Patient Receiving Adalimumab for Idiopathic Uveitis.Turk J Ophthalmol. 2024 Apr 19;54(2):112-115. doi: 10.4274/tjo.galenos.2024.04575. Turk J Ophthalmol. 2024. PMID: 38646109 Free PMC article.
References
-
- Exarchou SA, Voulgari PV, Markatseli TE, Zioga A, Drosos AA. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328–331. - PubMed
-
- Smith DI, Heffernan MP. Vitiligo after the resolution of psoriatic plaques during treatment with adalimumab. J Am Acad Dermatol. 2008;58(2 suppl):S50–S52. - PubMed
-
- Ramírez-Hernández M, Marras C, Martínez-Escribano JA. Infliximab-induced vitiligo. Dermatology. 2005;210:79–80. - PubMed
-
- Lv Y, Li Q, Wang L, Gao T. Use of anti-tumor necrosis factor agents: a possible therapy for vitiligo. Med Hypotheses. 2009;72:546–547. - PubMed
Publication types
LinkOut - more resources
Full Text Sources