Minimal antizyme peptide fully functioning in the binding and inhibition of ornithine decarboxylase and antizyme inhibitor
- PMID: 21931692
- PMCID: PMC3170320
- DOI: 10.1371/journal.pone.0024366
Minimal antizyme peptide fully functioning in the binding and inhibition of ornithine decarboxylase and antizyme inhibitor
Abstract
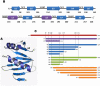
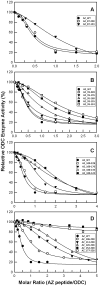
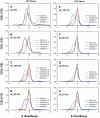
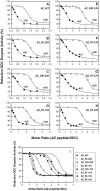
Antizyme (AZ) is a protein with 228 amino acid residues that regulates ornithine decarboxylase (ODC) by binding to ODC and dissociating its homodimer, thus inhibiting its enzyme activity. Antizyme inhibitor (AZI) is homologous to ODC, but has a higher affinity than ODC for AZ. In this study, we quantified the biomolecular interactions between AZ and ODC as well as AZ and AZI to identify functional AZ peptides that could bind to ODC and AZI and inhibit their function as efficiently as the full-length AZ protein. For these AZ peptides, the inhibitory ability of AZ_95-228 was similar to that of AZ_WT. Furthermore, AZ_95-176 displayed an inhibition (IC(50): 0.20 µM) similar to that of AZ-95-228 (IC(50): 0.16 µM), even though a large segment spanning residues 177-228 was deleted. However, further deletion of AZ_95-176 from either the N-terminus or the C-terminus decreased its ability to inhibit ODC. The AZ_100-176 and AZ_95-169 peptides displayed a noteworthy decrease in ability to inhibit ODC, with IC(50) values of 0.43 and 0.37 µM, respectively. The AZ_95-228, AZ_100-228 and AZ_95-176 peptides had IC(50) values comparable to that of AZ_WT and formed AZ-ODC complexes with K(d,AZ-ODC) values of 1.5, 5.3 and 5.6 µM, respectively. Importantly, our data also indicate that AZI can rescue AZ peptide-inhibited ODC enzyme activity and that it can bind to AZ peptides with a higher affinity than ODC. Together, these data suggest that these truncated AZ proteins retain their AZI-binding ability. Thus, we suggest that AZ_95-176 is the minimal AZ peptide that is fully functioning in the binding of ODC and AZI and inhibition of their function.
Conflict of interest statement
Figures
References
-
- Coffino P. Regulation of cellular polyamines by antizyme. Nat Rev Mol Cell Biol. 2001;2:188–194. - PubMed
-
- Pegg AE, McCann PP. Polyamine metabolism and function. Am J Physiol. 1982;243:C212–221. - PubMed
-
- Tabor CW, Tabor H. Polyamines. Annu Rev Biochem. 1984;53:749–790. - PubMed
-
- Marton LJ, Pegg AE. Polyamines as targets for therapeutic intervention. Annu Rev Pharmacol Toxicol. 1995;35:55–91. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

