The locus coeruleus is directly implicated in L-DOPA-induced dyskinesia in parkinsonian rats: an electrophysiological and behavioural study
- PMID: 21931808
- PMCID: PMC3170382
- DOI: 10.1371/journal.pone.0024679
The locus coeruleus is directly implicated in L-DOPA-induced dyskinesia in parkinsonian rats: an electrophysiological and behavioural study
Abstract
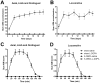
Despite being the most effective treatment for Parkinson's disease, L-DOPA causes a development of dyskinetic movements in the majority of treated patients. L-DOPA-induced dyskinesia is attributed to a dysregulated dopamine transmission within the basal ganglia, but serotonergic and noradrenergic systems are believed to play an important modulatory role. In this study, we have addressed the role of the locus coeruleus nucleus (LC) in a rat model of L-DOPA-induced dyskinesia. Single-unit extracellular recordings in vivo and behavioural and immunohistochemical approaches were applied in rats rendered dyskinetic by the destruction of the nigrostriatal dopamine neurons followed by chronic treatment with L-DOPA. The results showed that L-DOPA treatment reversed the change induced by 6-hydroxydopamine lesions on LC neuronal activity. The severity of the abnormal involuntary movements induced by L-DOPA correlated with the basal firing parameters of LC neuronal activity. Systemic administration of the LC-selective noradrenergic neurotoxin N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine did not modify axial, limb, and orolingual dyskinesia, whereas chemical destruction of the LC with ibotenic acid significantly increased the abnormal involuntary movement scores. These results are the first to demonstrate altered LC neuronal activity in 6-OHDA lesioned rats treated with L-DOPA, and indicate that an intact noradrenergic system may limit the severity of this movement disorder.
Conflict of interest statement
Figures

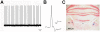
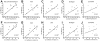
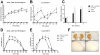
Similar articles
-
Behavioral and neurochemical effects of noradrenergic depletions with N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine in 6-hydroxydopamine-induced rat model of Parkinson's disease.Behav Brain Res. 2004 May 5;151(1-2):191-9. doi: 10.1016/j.bbr.2003.08.016. Behav Brain Res. 2004. PMID: 15084435
-
Bilateral subthalamic nucleus lesion reverses L-dopa-induced motor fluctuations and facilitates dyskinetic movements in hemiparkinsonian rats.Synapse. 2004 Feb;51(2):140-50. doi: 10.1002/syn.10291. Synapse. 2004. PMID: 14618681
-
Acute L-DOPA administration reverses changes in firing pattern and low frequency oscillatory activity in the entopeduncular nucleus from long term L-DOPA treated 6-OHDA-lesioned rats.Exp Neurol. 2019 Dec;322:113036. doi: 10.1016/j.expneurol.2019.113036. Epub 2019 Aug 16. Exp Neurol. 2019. PMID: 31425688
-
L-DOPA-induced dyskinesia in adult rats with a unilateral 6-OHDA lesion of dopamine neurons is paralleled by increased c-fos gene expression in the subthalamic nucleus.Eur J Neurosci. 2006 May;23(9):2395-403. doi: 10.1111/j.1460-9568.2006.04758.x. Eur J Neurosci. 2006. PMID: 16706847
-
mGlu5, Dopamine D2 and Adenosine A2A Receptors in L-DOPA-induced Dyskinesias.Curr Neuropharmacol. 2016;14(5):481-93. doi: 10.2174/1570159x14666151201185652. Curr Neuropharmacol. 2016. PMID: 26639458 Free PMC article. Review.
Cited by
-
Modulation of L-DOPA's antiparkinsonian and dyskinetic effects by α2-noradrenergic receptors within the locus coeruleus.Neuropharmacology. 2015 Aug;95:215-25. doi: 10.1016/j.neuropharm.2015.03.008. Epub 2015 Mar 25. Neuropharmacology. 2015. PMID: 25817388 Free PMC article.
-
The role of the subthalamic nucleus in L-DOPA induced dyskinesia in 6-hydroxydopamine lesioned rats.PLoS One. 2012;7(8):e42652. doi: 10.1371/journal.pone.0042652. Epub 2012 Aug 6. PLoS One. 2012. PMID: 22880070 Free PMC article.
-
Presynaptic Mechanisms of l-DOPA-Induced Dyskinesia: The Findings, the Debate, and the Therapeutic Implications.Front Neurol. 2014 Dec 15;5:242. doi: 10.3389/fneur.2014.00242. eCollection 2014. Front Neurol. 2014. PMID: 25566170 Free PMC article. Review.
-
Modulation of the subthalamic nucleus activity by serotonergic agents and fluoxetine administration.Psychopharmacology (Berl). 2014 May;231(9):1913-24. doi: 10.1007/s00213-013-3333-0. Epub 2013 Nov 24. Psychopharmacology (Berl). 2014. PMID: 24271033 Free PMC article.
-
Changes in Day/Night Activity in the 6-OHDA-Induced Experimental Model of Parkinson's Disease: Exploring Prodromal Biomarkers.Front Neurosci. 2020 Oct 14;14:590029. doi: 10.3389/fnins.2020.590029. eCollection 2020. Front Neurosci. 2020. PMID: 33154717 Free PMC article.
References
-
- Samii A, Nutt JG, Ransom BR. Parkinson's disease. Lancet. 2004;363:1783–1793. - PubMed
-
- Kish SJ, Shannak K, Hornykiewicz O. Uneven pattern of dopamine loss in the striatum of patients with idiopathic Parkinson's disease. Pathophysiologic and clinical implications. N Engl J Med. 1988;318:876–880. - PubMed
-
- Ahlskog JE, Muenter MD. Frequency of levodopa-related dyskinesias and motor fluctuations as estimated from the cumulative literature. Mov Disord. 2001;16:448–458. - PubMed
-
- Berthet A, Bezard E. Dopamine receptors and L-dopa-induced dyskinesia. Parkinsonism Relat Disord. 2009;15(Suppl 4):S8–12. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

