The NOTCH pathway contributes to cell fate decision in myelopoiesis
- PMID: 21933862
- PMCID: PMC3232256
- DOI: 10.3324/haematol.2011.044115
The NOTCH pathway contributes to cell fate decision in myelopoiesis
Abstract
Background: Controversy persists regarding the role of Notch signaling in myelopoiesis. We have used genetic approaches, employing two Notch zebrafish mutants deadly seven (DES) and beamter (BEA) with disrupted function of notch1a and deltaC, respectively, and Notch1a morphants to analyze the development of leukocyte populations in embryonic and mature fish.
Design and methods: Myelomonocytes were quantified in early embryos by in situ hybridization using a myeloper-oxidase (mpx) probe. Morpholinos were used to knock down expression of Notch1a or DeltaC. Wound healing assays and/or flow cytometry were used to quantify myelomonocytes in 5-day post-fertilization (dpf) Notch mutants (BEA and DES), morphants or pu.1:GFP, mpx:GFP and fms:RFP transgenic embryos. Flow cytometry was performed on 2-3 month old mutant fish.
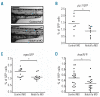
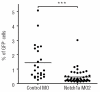
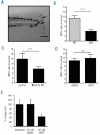
Results: The number of mpx(+) cells in embryos was reduced at 48 hpf (but not at 26 hpf) in DES compared to WT. At 5 dpf this was reflected by a reduction in the number of myelomonocytic cells found at the wound site in mutants and in Notch1a morphants. This was due to a reduced number of myelomonocytes developing rather than a deficit in the migratory ability since transient inhibition of Notch signaling using DAPT had no effect. The early deficit in myelopoiesis was maintained into later life, 2-3 month old BEA and DES fish having a decreased proportion of myelomonocytes in both the hematopoietic organ (kidney marrow) and the periphery (coelomic cavity).
Conclusions: Our results indicate that defects in Notch signaling affect definitive hematopoiesis, altering myelopoiesis from the early stages of development into the adult.
Figures



Comment in
-
The Notch signaling pathway in hematopoiesis and hematologic malignancies.Haematologica. 2011 Dec;96(12):1735-7. doi: 10.3324/haematol.2011.055954. Haematologica. 2011. PMID: 22147769 Free PMC article. No abstract available.
Similar articles
-
The Notch signaling pathway in hematopoiesis and hematologic malignancies.Haematologica. 2011 Dec;96(12):1735-7. doi: 10.3324/haematol.2011.055954. Haematologica. 2011. PMID: 22147769 Free PMC article. No abstract available.
-
Involvement of Delta/Notch signaling in zebrafish adult pigment stripe patterning.Development. 2014 Jan;141(2):318-24. doi: 10.1242/dev.099804. Epub 2013 Dec 4. Development. 2014. PMID: 24306107 Free PMC article.
-
Different combinations of Notch ligands and receptors regulate V2 interneuron progenitor proliferation and V2a/V2b cell fate determination.Dev Biol. 2014 Jul 15;391(2):196-206. doi: 10.1016/j.ydbio.2014.04.011. Epub 2014 Apr 24. Dev Biol. 2014. PMID: 24768892
-
Delta/Jagged-mediated Notch signaling induces the differentiation of agr2-positive epidermal mucous cells in zebrafish embryos.PLoS Genet. 2021 Dec 28;17(12):e1009969. doi: 10.1371/journal.pgen.1009969. eCollection 2021 Dec. PLoS Genet. 2021. PMID: 34962934 Free PMC article.
-
Notch signaling controls the differentiation of transporting epithelia and multiciliated cells in the zebrafish pronephros.Development. 2007 Mar;134(6):1111-22. doi: 10.1242/dev.02806. Epub 2007 Feb 7. Development. 2007. PMID: 17287248
Cited by
-
Hyperglycemia inhibits osteoblastogenesis of rat bone marrow stromal cells via activation of the Notch2 signaling pathway.Int J Med Sci. 2019 May 10;16(5):696-703. doi: 10.7150/ijms.32707. eCollection 2019. Int J Med Sci. 2019. PMID: 31217737 Free PMC article.
-
The Notch signaling pathway in hematopoiesis and hematologic malignancies.Haematologica. 2011 Dec;96(12):1735-7. doi: 10.3324/haematol.2011.055954. Haematologica. 2011. PMID: 22147769 Free PMC article. No abstract available.
-
Targeting oncogenic Notch signaling with SERCA inhibitors.J Hematol Oncol. 2021 Jan 6;14(1):8. doi: 10.1186/s13045-020-01015-9. J Hematol Oncol. 2021. PMID: 33407740 Free PMC article. Review.
-
The biochemistry of hematopoietic stem cell development.Biochim Biophys Acta. 2013 Feb;1830(2):2395-403. doi: 10.1016/j.bbagen.2012.10.004. Epub 2012 Oct 12. Biochim Biophys Acta. 2013. PMID: 23069720 Free PMC article. Review.
-
Notch signaling: its roles and therapeutic potential in hematological malignancies.Oncotarget. 2016 May 17;7(20):29804-23. doi: 10.18632/oncotarget.7772. Oncotarget. 2016. PMID: 26934331 Free PMC article. Review.
References
-
- Yuan JS, Kousis PC, Suliman S, Visan I, Guidos CJ. Functions of notch signaling in the immune system: consensus and controversies. Annu Rev Immunol. 2010;23:343–65. - PubMed
-
- Ohishi K, Katayama N, Shiku H, Varnum-Finney B, Bernstein ID. Notch signalling in hematopoiesis. Semin Cell Dev Biol. 2003;14(2):143–50. - PubMed
-
- Jarriault S, Brou C, Logeat F, Schroeter EH, Kopan R, Israel A. Signalling downstream of activated mammalian Notch. Nature. 1995;377(6547):355–8. - PubMed
-
- Ellisen LW, Bird J, West DC, Soreng AL, Reynolds TC, Smith SD, Sklar J. TAN-1, the human homolog of the Drosophila notch gene, is broken by chromosomal transloca-tions in T lymphoblastic neoplasms. Cell. 1991;66:649–61. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

