A systems approach for tumor pharmacokinetics
- PMID: 21935441
- PMCID: PMC3173470
- DOI: 10.1371/journal.pone.0024696
A systems approach for tumor pharmacokinetics
Abstract
Recent advances in genome inspired target discovery, small molecule screens, development of biological and nanotechnology have led to the introduction of a myriad of new differently sized agents into the clinic. The differences in small and large molecule delivery are becoming increasingly important in combination therapies as well as the use of drugs that modify the physiology of tumors such as anti-angiogenic treatment. The complexity of targeting has led to the development of mathematical models to facilitate understanding, but unfortunately, these studies are often only applicable to a particular molecule, making pharmacokinetic comparisons difficult. Here we develop and describe a framework for categorizing primary pharmacokinetics of drugs in tumors. For modeling purposes, we define drugs not by their mechanism of action but rather their rate-limiting step of delivery. Our simulations account for variations in perfusion, vascularization, interstitial transport, and non-linear local binding and metabolism. Based on a comparison of the fundamental rates determining uptake, drugs were classified into four categories depending on whether uptake is limited by blood flow, extravasation, interstitial diffusion, or local binding and metabolism. Simulations comparing small molecule versus macromolecular drugs show a sharp difference in distribution, which has implications for multi-drug therapies. The tissue-level distribution differs widely in tumors for small molecules versus macromolecular biologic drugs, and this should be considered in the design of agents and treatments. An example using antibodies in mouse xenografts illustrates the different in vivo behavior. This type of transport analysis can be used to aid in model development, experimental data analysis, and imaging and therapeutic agent design.
Conflict of interest statement
Figures


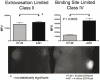
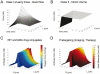
Similar articles
-
Multiscale Modeling of Antibody-Drug Conjugates: Connecting Tissue and Cellular Distribution to Whole Animal Pharmacokinetics and Potential Implications for Efficacy.AAPS J. 2016 Sep;18(5):1117-1130. doi: 10.1208/s12248-016-9940-z. Epub 2016 Jun 10. AAPS J. 2016. PMID: 27287046 Free PMC article.
-
Predictive pharmacokinetic-pharmacodynamic modeling of tumor growth after administration of an anti-angiogenic agent, bevacizumab, as single-agent and combination therapy in tumor xenografts.Cancer Chemother Pharmacol. 2013 May;71(5):1147-57. doi: 10.1007/s00280-013-2107-z. Epub 2013 Feb 21. Cancer Chemother Pharmacol. 2013. PMID: 23430120
-
Antitumor activity of targeted and cytotoxic agents in murine subcutaneous tumor models correlates with clinical response.Clin Cancer Res. 2012 Jul 15;18(14):3846-55. doi: 10.1158/1078-0432.CCR-12-0738. Epub 2012 May 30. Clin Cancer Res. 2012. PMID: 22648270
-
Image-guided mathematical modeling for pharmacological evaluation of nanomaterials and monoclonal antibodies.Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2020 Sep;12(5):e1628. doi: 10.1002/wnan.1628. Epub 2020 Apr 21. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2020. PMID: 32314552 Free PMC article. Review.
-
Metabolism and Distribution of Novel Tumor Targeting Drugs In Vivo.Curr Drug Metab. 2020;21(13):996-1008. doi: 10.2174/1389200221666201112110638. Curr Drug Metab. 2020. PMID: 33183197
Cited by
-
Discovery, affinity maturation and multimerization of small molecule ligands against human tyrosinase and tyrosinase-related protein 1.RSC Med Chem. 2020 Nov 13;12(3):363-369. doi: 10.1039/d0md00310g. RSC Med Chem. 2020. PMID: 34041485 Free PMC article.
-
Prediction of drug disposition on the basis of its chemical structure.Clin Pharmacokinet. 2013 Jun;52(6):415-31. doi: 10.1007/s40262-013-0042-0. Clin Pharmacokinet. 2013. PMID: 23463353 Review.
-
The effect of ligand amount, affinity and internalization on PSMA-targeted imaging and therapy: A simulation study using a PBPK model.Sci Rep. 2019 Dec 27;9(1):20041. doi: 10.1038/s41598-019-56603-8. Sci Rep. 2019. PMID: 31882829 Free PMC article.
-
Multichannel imaging to quantify four classes of pharmacokinetic distribution in tumors.J Pharm Sci. 2014 Oct;103(10):3276-86. doi: 10.1002/jps.24086. Epub 2014 Jul 21. J Pharm Sci. 2014. PMID: 25048378 Free PMC article.
-
Population Pharmacokinetics of Tamibarotene in Pediatric and Young Adult Patients with Recurrent or Refractory Solid Tumors.Curr Oncol. 2024 Nov 14;31(11):7155-7164. doi: 10.3390/curroncol31110527. Curr Oncol. 2024. PMID: 39590158 Free PMC article. Clinical Trial.
References
-
- Minchinton AI, Tannock IF. Drug penetration in solid tumours. Nature Reviews Cancer. 2006;6:583–592. - PubMed
-
- van der Greef J, McBurney RN. Innovation - Rescuing drug discovery: in vivo systems pathology and systems pharmacology. Nature Reviews Drug Discovery. 2005;4:961–967. - PubMed
-
- Tolmachev V, Wallberg H, Sandstrom M, Hansson M, Wennborg A, et al. Optimal specific radioactivity of anti-HER2 Affibody molecules enables discrimination between xenografts with high and low HER2 expression levels. Eur J Nucl Med Mol Imaging. 2011. - PubMed
-
- Allerheiligen SRB. Next-Generation Model-Based Drug Discovery and Development: Quantitative and Systems Pharmacology. Clinical Pharmacology & Therapeutics. 2010;88:135–137. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

