Review of Reported Clinical Information System Adverse Events in US Food and Drug Administration Databases
- PMID: 21938265
- PMCID: PMC3175794
- DOI: 10.4338/ACI-2010-11-RA-0064
Review of Reported Clinical Information System Adverse Events in US Food and Drug Administration Databases
Abstract
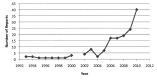
BACKGROUND: The US FDA has been collecting information on medical devices involved in significant adverse advents since 1984. These reports have been used by researchers to advise clinicians on potential risks and complications of using these devices. OBJECTIVE: Research adverse events related to the use of Clinical Information Systems (CIS) as reported in FDA databases. METHODS: Three large, national, adverse event medical device databases were examined for reports pertaining to CIS. RESULTS: One hundred and twenty unique reports (from over 1.4 million reports) were found, representing 32 manufacturers. The manifestations of these adverse events included: missing or incorrect data, data displayed for the wrong patient, chaos during system downtime and system unavailable for use. Analysis of these reports illustrated events associated with system design, implementation, use, and support. CONCLUSION: The identified causes can be used by manufacturers to improve their products and by clinical facilities and providers to adjust their workflow and implementation processes appropriately. The small number of reports found indicates a need to raise awareness regarding publicly available tools for documenting problems with CIS and for additional reporting and dialog between manufacturers, organizations, and users.
Figures
Similar articles
-
Late adverse event reporting from medical device manufacturers to the US Food and Drug Administration: cross sectional study.BMJ. 2025 Mar 12;388:e081518. doi: 10.1136/bmj-2024-081518. BMJ. 2025. PMID: 40081838 Free PMC article.
-
Folic acid supplementation and malaria susceptibility and severity among people taking antifolate antimalarial drugs in endemic areas.Cochrane Database Syst Rev. 2022 Feb 1;2(2022):CD014217. doi: 10.1002/14651858.CD014217. Cochrane Database Syst Rev. 2022. PMID: 36321557 Free PMC article.
-
The future of Cochrane Neonatal.Early Hum Dev. 2020 Nov;150:105191. doi: 10.1016/j.earlhumdev.2020.105191. Epub 2020 Sep 12. Early Hum Dev. 2020. PMID: 33036834
-
An Analysis of the FDA MAUDE Database and the Search for Cobalt Toxicity in Class 3 Johnson & Johnson/DePuy Metal-on-Metal Hip Implants.J Patient Saf. 2018 Dec;14(4):e89-e96. doi: 10.1097/PTS.0000000000000534. J Patient Saf. 2018. PMID: 30308590
-
Trocar-associated injuries and fatalities: an analysis of 1399 reports to the FDA.J Minim Invasive Gynecol. 2005 Jul-Aug;12(4):302-7. doi: 10.1016/j.jmig.2005.05.008. J Minim Invasive Gynecol. 2005. PMID: 16036187 Review.
Cited by
-
A systematic review of trials evaluating success factors of interventions with computerised clinical decision support.Implement Sci. 2018 Aug 20;13(1):114. doi: 10.1186/s13012-018-0790-1. Implement Sci. 2018. PMID: 30126421 Free PMC article.
-
Who Watches the Watchers. Working Towards Safety for EHR Knowledge Resources.Appl Clin Inform. 2017 Jun 28;8(2):680-685. doi: 10.4338/ACI-2017-02-IE-0032. Appl Clin Inform. 2017. PMID: 28657638 Free PMC article.
-
Emergency Department Workload and Crowding During a Major Electronic Health Record Breakdown.Front Public Health. 2019 Sep 12;7:267. doi: 10.3389/fpubh.2019.00267. eCollection 2019. Front Public Health. 2019. PMID: 31572707 Free PMC article.
-
Exploring the sociotechnical intersection of patient safety and electronic health record implementation.J Am Med Inform Assoc. 2014 Feb;21(e1):e28-34. doi: 10.1136/amiajnl-2013-001762. Epub 2013 Sep 19. J Am Med Inform Assoc. 2014. PMID: 24052536 Free PMC article.
-
Creating an oversight infrastructure for electronic health record-related patient safety hazards.J Patient Saf. 2011 Dec;7(4):169-74. doi: 10.1097/PTS.0b013e31823d8df0. J Patient Saf. 2011. PMID: 22080284 Free PMC article.
References
-
- NPR Staff Anti-virus program update wreaks havoc with PCs: NPR [Internet] 2010Apr 21 [cited 2010 Apr 25];Available from: http://www.npr.org/templates/story/story.php?storyId=126168997&sc=17&f=1001
-
- Dearne K. Medicare glitch affects records. The Australian [Internet] 2010Apr 20 [cited 2010 Apr 25];Available from: http://www.theaustralian.com.au/australian-it/medicare-glitch-affects-re...
-
- MAUDE – Manufacturer and User Facility Device Experience [Internet] [cited 2010 Apr 15]; Available from: http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfMAUDE/search.CFM
-
- Koppel R, Kreda D. Health care information technology vendors' "hold harm-less" clause: implications for patients and clinicians. JAMA 2009; 301(12): 1276-1278 - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources