sRNA-Xcc1, an integron-encoded transposon- and plasmid-transferred trans-acting sRNA, is under the positive control of the key virulence regulators HrpG and HrpX of Xanthomonas campestris pathovar campestris
- PMID: 21941121
- PMCID: PMC3256417
- DOI: 10.4161/rna.8.6.16690
sRNA-Xcc1, an integron-encoded transposon- and plasmid-transferred trans-acting sRNA, is under the positive control of the key virulence regulators HrpG and HrpX of Xanthomonas campestris pathovar campestris
Abstract
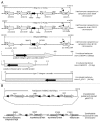
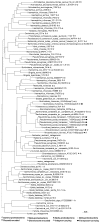
sRNA-Xcc1 is a trans-acting sRNA recently identified from the plant pathogenic bacterium Xanthomonas campestris pathovar campestris (Xcc). Here, the phylogenetic distribution, predicted secondary structure and regulation of expression of sRNA-Xcc1 were analyzed. The analysis showed (1) a total 81 sRNA-Xcc1 homologs that are found in some bacterial strains that are taxonomically unrelated, belonging to the α-, β-, γ- and δ-proteobacteria (2) that some sRNA-Xcc1 homologs are located in a plasmid-borne transposon or near a transposase coding gene, (3) that sRNA-Xcc1 is encoded by a integron gene cassette in Xcc and sRNA-Xcc1 homologs occur in integron gene cassettes of some uncultured bacteria and (4) that sRNA-Xcc1 homologs have a highly conserved sequence motif and a stable consensus secondary structure. These findings strongly support the idea that sRNA-Xcc1 represents a novel family of sRNAs which may be originally captured by integrons from natural environments and then spread among different bacterial species via horizontal gene transfer, possibly by means of transposons and plasmids. The expression analysis results demonstrated that the transcription of sRNA-Xcc1 is under the positive control of the key virulence regulators HrpG and HrpX, indicating that sRNA-Xcc1 may be involved in the virulence regulation of Xcc.
Figures

References
-
- Hayward AC. The host of Xanthomonas. In: Swings JG, Civerolo EL, editors. Xanthomonas. London: Chapman and Hall; 1993. pp. 51–54.
-
- da Silva AC, Ferro JA, Reinach FC, Farah CS, Furlan LR, Quaggio RB, et al. Comparison of the genomes of two Xanthomonas pathogens with differing host specificities. Nature. 2002;417:459–463. - PubMed
-
- Vorhölter FJ, Schneiker S, Goesmann A, Krause L, Bekel T, Kaiser O, et al. The genome of Xanthomonas campestris pv. campestris B100 and its use for the reconstruction of metabolic pathways involved in xanthan biosynthesis. J Biotechnol. 2008;134:33–45. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
