Silver ions disrupt K⁺ homeostasis and cellular integrity in intact barley (Hordeum vulgare L.) roots
- PMID: 21948852
- PMCID: PMC3245464
- DOI: 10.1093/jxb/err267
Silver ions disrupt K⁺ homeostasis and cellular integrity in intact barley (Hordeum vulgare L.) roots
Abstract
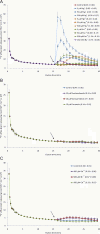
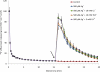
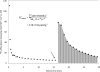
The heavy metals silver, gold, and mercury can strongly inhibit aquaporin-mediated water flow across plant cell membranes, but critical examinations of their side effects are rare. Here, the short-lived radiotracer (42)K is used to demonstrate that these metals, especially silver, profoundly change potassium homeostasis in roots of intact barley (Hordeum vulgare L.) plants, by altering unidirectional K(+) fluxes. Doses as low as 5 μM AgNO(3) rapidly reduced K(+) influx to 5% that of controls, and brought about pronounced and immediate increases in K(+) efflux, while higher doses of Au(3+) and Hg(2+) were required to produce similar responses. Reduced influx and enhanced efflux of K(+) resulted in a net loss of >40% of root tissue K(+) during a 15 min application of 500 μM AgNO(3), comprising the entire cytosolic potassium pool and about a third of the vacuolar pool. Silver also brought about major losses of UV-absorbing compounds, total electrolytes, and NH(4)(+). Co-application, with silver, of the channel blockers Cs(+), TEA(+), or Ca(2+), did not affect the enhanced efflux, ruling out the involvement of outwardly rectifying ion channels. Taken together with an examination of propidium iodide staining under confocal microscopy, the results indicate that silver ions affect K(+) homeostasis by directly inhibiting K(+) influx at lower concentrations, and indirectly inhibiting K(+) influx and enhancing K(+) efflux, via membrane destruction, at higher concentrations. Ni(2+), Cd(2+), and Pb(2+), three heavy metals not generally known to affect aquaporins, did not enhance K(+) efflux or cause propidium iodide incorporation. The study reveals strong and previously unknown effects of major aquaporin inhibitors and recommends caution in their application.
Figures



Similar articles
-
42K analysis of sodium-induced potassium efflux in barley: mechanism and relevance to salt tolerance.New Phytol. 2010 Apr;186(2):373-84. doi: 10.1111/j.1469-8137.2009.03169.x. Epub 2010 Jan 28. New Phytol. 2010. PMID: 20122133
-
Rapid, futile K+ cycling and pool-size dynamics define low-affinity potassium transport in barley.Plant Physiol. 2006 Aug;141(4):1494-507. doi: 10.1104/pp.106.082701. Epub 2006 Jun 30. Plant Physiol. 2006. PMID: 16815955 Free PMC article.
-
Cytosolic potassium homeostasis revisited: 42K-tracer analysis in Hordeum vulgare L. reveals set-point variations in [K+].Planta. 2003 Aug;217(4):540-6. doi: 10.1007/s00425-003-1032-5. Epub 2003 May 1. Planta. 2003. PMID: 12728317
-
Regulation and mechanism of potassium release from barley roots: an in planta 42K+ analysis.New Phytol. 2010 Dec;188(4):1028-38. doi: 10.1111/j.1469-8137.2010.03436.x. Epub 2010 Aug 20. New Phytol. 2010. PMID: 20731780
-
Complexity of potassium acquisition: how much flows through channels?Plant Signal Behav. 2013 Jul;8(7):e24799. doi: 10.4161/psb.24799. Epub 2013 Jul 1. Plant Signal Behav. 2013. PMID: 23656868 Free PMC article.
Cited by
-
The Role of Silicon in Higher Plants under Salinity and Drought Stress.Front Plant Sci. 2016 Jul 18;7:1072. doi: 10.3389/fpls.2016.01072. eCollection 2016. Front Plant Sci. 2016. PMID: 27486474 Free PMC article. Review.
-
Capacity and plasticity of potassium channels and high-affinity transporters in roots of barley and Arabidopsis.Plant Physiol. 2013 May;162(1):496-511. doi: 10.1104/pp.113.215913. Epub 2013 Apr 3. Plant Physiol. 2013. PMID: 23553635 Free PMC article.
-
Argovit™ Silver Nanoparticles Effects on Allium cepa: Plant Growth Promotion without Cyto Genotoxic Damage.Nanomaterials (Basel). 2020 Jul 16;10(7):1386. doi: 10.3390/nano10071386. Nanomaterials (Basel). 2020. PMID: 32708646 Free PMC article.
-
Uptake and bioaccumulation of iron oxide nanoparticles (Fe3O4) in barley (Hordeum vulgare L.): effect of particle-size.Environ Sci Pollut Res Int. 2024 Mar;31(14):22171-22186. doi: 10.1007/s11356-024-32378-y. Epub 2024 Feb 26. Environ Sci Pollut Res Int. 2024. PMID: 38403831
-
Hydraulic conductivity and contribution of aquaporins to water uptake in roots of four sunflower genotypes.Bot Stud. 2014 Dec;55(1):75. doi: 10.1186/s40529-014-0075-1. Epub 2014 Oct 30. Bot Stud. 2014. PMID: 28510954 Free PMC article.
References
-
- Agre P, Lee MD, Devidas S, Guggino WB. Aquaporins and ion conductance. Science. 1997;275:1490. - PubMed
-
- Al-Khayri JM, Al-Bahrany AM. Silver nitrate and 2-isopentyladenine promote somatic embryogenesis in date palm (Phoenix dactylifera L.) Scientia Horticulturae. 2001;89:291–298.
-
- Asharani PV, Sethu S, Vadukumpully S, Zhong SP, Lim CT, Hande MP, Valiyaveettil S. Investigations on the structural damage in human erythrocytes exposed to silver, gold, and platinum nanoparticles. Advanced Functional Materials. 2010;20:1233–1242.
-
- Balkos KD, Britto DT, Kronzucker HJ. Optimization of ammonium acquisition and metabolism by potassium in rice (Oryza sativa L. cv. IR-72) Plant, Cell and Environment. 2010;33:23–34. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

