Inhibitory interneurons in a cortical column form hot zones of inhibition in layers 2 and 5A
- PMID: 21949377
- PMCID: PMC3189020
- DOI: 10.1073/pnas.1113648108
Inhibitory interneurons in a cortical column form hot zones of inhibition in layers 2 and 5A
Abstract
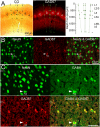
Although physiological data on microcircuits involving a few inhibitory neurons in the mammalian cerebral cortex are available, data on the quantitative relation between inhibition and excitation in cortical circuits involving thousands of neurons are largely missing. Because the distribution of neurons is very inhomogeneous in the cerebral cortex, it is critical to map all neurons in a given volume rather than to rely on sparse sampling methods. Here, we report the comprehensive mapping of interneurons (INs) in cortical columns of rat somatosensory cortex, immunolabeled for neuron-specific nuclear protein and glutamate decarboxylase. We found that a column contains ~2,200 INs (11.5% of ~19,000 neurons), almost a factor of 2 less than previously estimated. The density of GABAergic neurons was inhomogeneous between layers, with peaks in the upper third of L2/3 and in L5A. IN density therefore defines a distinct layer 2 in the sensory neocortex. In addition, immunohistochemical markers of IN subtypes were layer-specific. The "hot zones" of inhibition in L2 and L5A match the reported low stimulus-evoked spiking rates of excitatory neurons in these layers, suggesting that these inhibitory hot zones substantially suppress activity in the neocortex.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Pouille F, Scanziani M. Enforcement of temporal fidelity in pyramidal cells by somatic feed-forward inhibition. Science. 2001;293:1159–1163. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

