Vibrational coherence spectroscopy of the heme domain in the CO-sensing transcriptional activator CooA
- PMID: 21961804
- PMCID: PMC3218251
- DOI: 10.1021/ja206152m
Vibrational coherence spectroscopy of the heme domain in the CO-sensing transcriptional activator CooA
Abstract
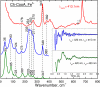
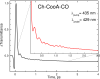
Femtosecond vibrational coherence spectroscopy was used to investigate the low-frequency vibrational dynamics of the heme in the carbon monoxide oxidation activator protein (CooA) from the thermophilic anaerobic bacterium Carboxydothermus hydrogenoformans (Ch-CooA). Low frequency vibrational modes are important because they are excited by the ambient thermal bath (k(B)T = 200 cm(-1)) and participate in thermally activated barrier crossing events. However, such modes are nearly impossible to detect in the aqueous phase using traditional spectroscopic methods. Here, we present the low frequency coherence spectra of the ferric, ferrous, and CO-bound forms of Ch-CooA in order to compare the protein-induced heme distortions in its active and inactive states. Distortions take place predominantly along the coordinates of low-frequency modes because of their weak force constants, and such distortions are reflected in the intensity of the vibrational coherence signals. A strong mode near ~90 cm(-1) in the ferrous form of Ch-CooA is suggested to contain a large component of heme ruffling, consistent with the imidazole-bound ferrous heme crystal structure, which shows a significant protein-induced heme distortion along this coordinate. A mode observed at ~228 cm(-1) in the six-coordinate ferrous state is proposed to be the ν(Fe-His) stretching vibration. The observation of the Fe-His mode indicates that photolysis of the N-terminal α-amino axial ligand takes place. This is followed by a rapid (~8.5 ps) transient absorption recovery, analogous to methionine rebinding in photolyzed ferrous cytochrome c. We have also studied CO photolysis in CooA, which revealed very strong photoproduct state coherent oscillations. The observation of heme-CO photoproduct oscillations is unusual because most other heme systems have CO rebinding kinetics that are too slow to make the measurement possible. The low frequency coherence spectrum of the CO-bound form of Ch-CooA shows a strong vibration at ~230 cm(-1) that is broadened and up-shifted compared to the ν(Fe-His) of Rr-CooA (216 cm(-1)). We propose that the stronger Fe-His bond is related to the enhanced thermal stability of Ch-CooA and that there is a smaller (time dependent) tilt of the histidine ring with respect to the heme plane in Ch-CooA. The appearance of strong modes at ~48 cm(-1) in both the ferrous and CO-bound forms of Ch-CooA is consistent with coupling of the heme doming distortion to the photolysis reaction in both samples. Upon CO binding and protein activation, a heme mode near 112 ± 5 cm(-1) disappears, probably indicating a decreased heme saddling distortion. This reflects changes in the heme environment and geometry that must be associated with the conformational transition activating the DNA-binding domain. Protein-specific DNA binding to the CO-bound form of Ch-CooA was also investigated, and although the CO rebinding kinetics are significantly perturbed, there are negligible changes in the low-frequency vibrational spectrum of the heme.
Figures







Similar articles
-
Effect of DNA binding on geminate CO recombination kinetics in CO-sensing transcription factor CooA.J Biol Chem. 2012 Jun 22;287(26):21729-40. doi: 10.1074/jbc.M112.345090. Epub 2012 Apr 28. J Biol Chem. 2012. PMID: 22544803 Free PMC article.
-
Crystal structure of CO-sensing transcription activator CooA bound to exogenous ligand imidazole.J Mol Biol. 2007 Mar 30;367(3):864-71. doi: 10.1016/j.jmb.2007.01.043. Epub 2007 Jan 23. J Mol Biol. 2007. PMID: 17292914
-
Identification of histidine 77 as the axial heme ligand of carbonmonoxy CooA by picosecond time-resolved resonance Raman spectroscopy.Biochemistry. 2000 Oct 24;39(42):12747-52. doi: 10.1021/bi0011476. Biochemistry. 2000. PMID: 11041838
-
CO sensing and regulation of gene expression by the transcriptional activator CooA.J Inorg Biochem. 2000 Nov;82(1-4):51-6. doi: 10.1016/s0162-0134(00)00139-2. J Inorg Biochem. 2000. PMID: 11132638 Review.
-
Biochemical and biophysical properties of the CO-sensing transcriptional activator CooA.Acc Chem Res. 2003 Nov;36(11):825-31. doi: 10.1021/ar020097p. Acc Chem Res. 2003. PMID: 14622029 Review.
Cited by
-
Ultrafast dynamics of heme distortion in the O2-sensor of a thermophilic anaerobe bacterium.Commun Chem. 2021 Mar 5;4(1):31. doi: 10.1038/s42004-021-00471-9. Commun Chem. 2021. PMID: 36697566 Free PMC article.
-
The diagnostic vibrational signature of pentacoordination in heme carbonyls.J Am Chem Soc. 2014 Jul 16;136(28):9818-21. doi: 10.1021/ja503191z. Epub 2014 Jul 7. J Am Chem Soc. 2014. PMID: 24950373 Free PMC article.
-
Effect of DNA binding on geminate CO recombination kinetics in CO-sensing transcription factor CooA.J Biol Chem. 2012 Jun 22;287(26):21729-40. doi: 10.1074/jbc.M112.345090. Epub 2012 Apr 28. J Biol Chem. 2012. PMID: 22544803 Free PMC article.
-
Ultrafast infrared spectroscopy reveals water-mediated coherent dynamics in an enzyme active site.Chem Sci. 2015 Jan 1;6(1):505-516. doi: 10.1039/c4sc02752c. Epub 2014 Oct 22. Chem Sci. 2015. PMID: 28936306 Free PMC article.
-
Characterizing Mode Anharmonicity and Huang-Rhys Factors Using Models of Femtosecond Coherence Spectra.J Phys Chem Lett. 2022 Jun 23;13(24):5413-5423. doi: 10.1021/acs.jpclett.1c04162. Epub 2022 Jun 9. J Phys Chem Lett. 2022. PMID: 35679146 Free PMC article.
References
-
- Vos MH. Biochim.Biophys.Acta. 2008;1777:15–31. - PubMed
-
- Cary SPL, Winger JA, Derbyshire ER, Marletta MA. Trends in Biochem.Sci. 2006;31:231–239. - PubMed
-
- Rodgers KR, Lukat-Rodgers GS. J.Inorg.Biochem. 2005;99:963–977. - PubMed
-
- Delgado-Nixon VM, Gonzalez G, Gilles-Gonzalez MA. Biochemistry. 2000;39:2685–2691. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

