5-lipoxygenase expression and tepoxalin-induced cell death in squamous cell carcinomas in cats
- PMID: 21962280
- PMCID: PMC10957241
- DOI: 10.2460/ajvr.72.10.1369
5-lipoxygenase expression and tepoxalin-induced cell death in squamous cell carcinomas in cats
Abstract
Objective: To assess expression pattern and subcellular compartmentalization of 5-lipoxygenase in cutaneous, UV radiation-induced, and oral squamous cell carcinomas (SCCs) in cats and determine the effects of cyclooxygenase or 5-lipoxygenase inhibition on proliferation or apoptosis in a feline oral squamous cell carcinoma (SCCF1) cell line.
Sample: 60 archived paraffin-embedded samples of SCCs from 60 cats and SCCF1 cells.
Procedures: Retrospective immunohistochemical analysis of the archived samples of SCCs (20 cutaneous, 20 UV radiation-induced, and 20 oral tumors) was performed. Cell culture proliferation assays involving SCCF1 cells were performed, and tepoxalin-induced apoptosis and signaling were examined via western blotting and annexin V staining.
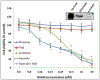
Results: Immunohistochemically, staining for 5-lipoxygenase was most frequently of greatest intensity in oral SCCs, whereas staining of cutaneous and UV radiation-induced lesions had less consistent 5-lipoxygenase expression. Exposure of SCCF1 cells to the 5-lipoxygenase inhibitor tepoxalin resulted in apoptosis; the effect appeared to be mediated via alteration of cell signaling rather than via suppression of lipid mediators that are typically produced as a result of 5-lipoxygenase activity.
Conclusions and clinical relevance: In cats, expression of 5-lipoxygenase in SCCs appeared to differ depending on tumor location. The influence of tepoxalin-induced 5-lipoxygenase inhibition on a 5-lipoxygenase-expressing cell line coupled with the notable expression of 5-lipoxygenase in oral SCCs suggested that 5-lipoxygenase inhibition may have therapeutic benefits in affected cats. Although the safety of tepoxalin in cats has yet to be investigated, 5-lipoxygenase inhibitors should be evaluated for use as a potential treatment for SCCs in that species.
Figures



References
-
- Vail DM, Withrow SJ. Tumors of the skin and subcutaneous tissues. In: Withrow SJ, Vail DM, eds. Small animal clinical oncology. 4th ed. St Louis: Saunders, 2007;375–401.
-
- Head KW, Else RW, Dubielzieg RR. Tumors of the alimentary tract. In: Meuten DJ, ed. Tumors in domestic animals. 4th ed. Ames, Iowa: Blackwell Publishing, 2002;401–482.
-
- Gil Z, Fiss DM. Contemporary management of head and neck tumors. Isr Med Assoc J 2009;11:296–300. - PubMed
-
- Pannone G, Bufo P, Grothey A, et al. Cyclooxygenase-2 expression in oral squamous cell carcinoma. Int J Immunopath Pharmacol 2004;17:273–282. - PubMed
-
- Pestili de Almeida EM, Piche C, Sirios J, et al. Expression of cyclo-oxygenase 2 in naturally occurring squamous cell carcinomas in dogs. J Histochem Cytochem 2001;49:867–875. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

