The calcineurin inhibitor tacrolimus activates the renal sodium chloride cotransporter to cause hypertension
- PMID: 21963515
- PMCID: PMC3192268
- DOI: 10.1038/nm.2497
The calcineurin inhibitor tacrolimus activates the renal sodium chloride cotransporter to cause hypertension
Abstract
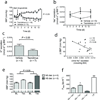
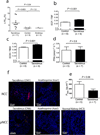
Calcineurin inhibitors (CNIs) are immunosuppressive drugs that are used widely to prevent rejection of transplanted organs and to treat autoimmune disease. Hypertension and renal tubule dysfunction, including hyperkalemia, hypercalciuria and acidosis, often complicate their use. These side effects resemble familial hyperkalemic hypertension, a genetic disease characterized by overactivity of the renal sodium chloride cotransporter (NCC) and caused by mutations in genes encoding WNK kinases. We hypothesized that CNIs induce hypertension by stimulating NCC. In wild-type mice, the CNI tacrolimus caused salt-sensitive hypertension and increased the abundance of phosphorylated NCC and the NCC-regulatory kinases WNK3, WNK4 and SPAK. We demonstrated the functional importance of NCC in this response by showing that tacrolimus did not affect blood pressure in NCC-knockout mice, whereas the hypertensive response to tacrolimus was exaggerated in mice overexpressing NCC. Moreover, hydrochlorothiazide, an NCC-blocking drug, reversed tacrolimus-induced hypertension. These observations were extended to humans by showing that kidney transplant recipients treated with tacrolimus had a greater fractional chloride excretion in response to bendroflumethiazide, another NCC-blocking drug, than individuals not treated with tacrolimus; renal NCC abundance was also greater. Together, these findings indicate that tacrolimus-induced chronic hypertension is mediated largely by NCC activation, and suggest that inexpensive and well-tolerated thiazide diuretics may be especially effective in preventing the complications of CNI treatment.
Figures


References
-
- Kim HC, et al. Primary immunosuppression with tacrolimus in kidney transplantation: three-year follow-up in a single center. Transplant. Proc. 2004;36:2082–2083. - PubMed
-
- Nijenhuis T, Hoenderop JG, Bindels RJ. Downregulation of Ca(2+) and Mg(2+) transport proteins in the kidney explains tacrolimus (FK506)-induced hypercalciuria and hypomagnesemia. J Am Soc Nephrol. 2004;15:549–557. - PubMed
-
- Mohebbi N, Mihailova M, Wagner CA. The calcineurin inhibitor FK506 (tacrolimus) is associated with transient metabolic acidosis and altered expression of renal acid-base transport proteins. American journal of physiology. Renal physiology. 2009;297:F499–F509. - PubMed
-
- Takeda Y, Miyamori I, Furukawa K, Inaba S, Mabuchi H. Mechanisms of FK 506-induced hypertension in the rat. Hypertension. 1999;33:130–136. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

