Identification of a novel selective serotonin reuptake inhibitor by coupling monoamine transporter-based virtual screening and rational molecular hybridization
- PMID: 21966587
- PMCID: PMC3181111
- DOI: 10.1021/cn200044x
Identification of a novel selective serotonin reuptake inhibitor by coupling monoamine transporter-based virtual screening and rational molecular hybridization
Abstract
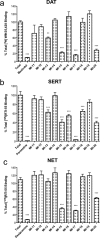
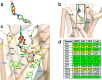
Ligand virtual screening (VS) using the vestibular binding pocket of a 3-D monoamine transporter (MAT) computational model followed by in vitro pharmacology led to the identification of a human serotonin transporter (hSERT) inhibitor with modest affinity (hSERT K(i) = 284 nM). Structural comparison of this VS-elucidated compound, denoted MI-17, to known SERT ligands led to the rational design and synthesis of DJLDU-3-79, a molecular hybrid of MI-17 and dual SERT/5-HT(1A) receptor antagonist SSA-426. Relative to MI-17, DJLDU-3-79 displayed 7-fold improvement in hSERT binding affinity and a 3-fold increase in [(3)H]-serotonin uptake inhibition potency at hSERT/HEK cells. This hybrid compound displayed a hSERT:hDAT selectivity ratio of 50:1, and a hSERT:hNET (human norepinephrine transporter) ratio of >200:1. In mice, DJLDU-3-79 decreased immobility in the tail suspension test comparable to the SSRI fluvoxamine, suggesting that DJLDU-3-79 may possess antidepressant properties. This proof of concept study highlights MAT virtual screening as a powerful tool for identifying novel inhibitor chemotypes and chemical fragments for rational inhibitor design.
Figures



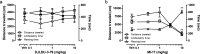
Similar articles
-
Rapid and sustained antidepressant properties of an NMDA antagonist/monoamine reuptake inhibitor identified via transporter-based virtual screening.Pharmacol Biochem Behav. 2016 Nov-Dec;150-151:22-30. doi: 10.1016/j.pbb.2016.08.007. Epub 2016 Aug 26. Pharmacol Biochem Behav. 2016. PMID: 27569602 Free PMC article.
-
Further studies on conformationally constrained tricyclic tropane analogues and their uptake inhibition at monoamine transporter sites: synthesis of (Z)-9-(substituted arylmethylene)-7-azatricyclo[4.3.1.0(3,7)]decanes as a novel class of serotonin transporter inhibitors.J Med Chem. 2002 Apr 25;45(9):1930-41. doi: 10.1021/jm0105373. J Med Chem. 2002. PMID: 11960503
-
Studies of the biogenic amine transporters. XI. Identification of a 1-[2-[bis(4-fluorophenyl)methoxy]ethyl]-4-(3-phenylpropyl)piperazine (GBR12909) analog that allosterically modulates the serotonin transporter.J Pharmacol Exp Ther. 2005 Aug;314(2):906-15. doi: 10.1124/jpet.105.084376. Epub 2005 Apr 28. J Pharmacol Exp Ther. 2005. PMID: 15860577
-
Discovery of novel-scaffold monoamine transporter ligands via in silico screening with the S1 pocket of the serotonin transporter.ACS Chem Neurosci. 2014 Sep 17;5(9):784-92. doi: 10.1021/cn500133b. Epub 2014 Jul 15. ACS Chem Neurosci. 2014. PMID: 25003748 Free PMC article. Review.
-
Y95 and E444 interaction required for high-affinity S-citalopram binding in the human serotonin transporter.ACS Chem Neurosci. 2011 Feb 16;2(2):75-81. doi: 10.1021/cn100066p. Epub 2010 Oct 27. ACS Chem Neurosci. 2011. PMID: 22778858 Free PMC article. Review.
Cited by
-
Designing modulators of monoamine transporters using virtual screening techniques.Front Pharmacol. 2015 Sep 29;6:223. doi: 10.3389/fphar.2015.00223. eCollection 2015. Front Pharmacol. 2015. PMID: 26483692 Free PMC article. Review.
-
Highly diastereoselective synthesis of tetrahydropyridines by a C-H activation-cyclization-reduction cascade.J Am Chem Soc. 2012 Mar 7;134(9):4064-7. doi: 10.1021/ja2119833. Epub 2012 Feb 22. J Am Chem Soc. 2012. PMID: 22356093 Free PMC article.
-
Discovery of novel selective serotonin reuptake inhibitors through development of a protein-based pharmacophore.J Chem Inf Model. 2011 Sep 26;51(9):2417-26. doi: 10.1021/ci200280m. Epub 2011 Sep 2. J Chem Inf Model. 2011. PMID: 21834587 Free PMC article.
-
Monoamine transporter structure, function, dynamics, and drug discovery: a computational perspective.AAPS J. 2012 Dec;14(4):820-31. doi: 10.1208/s12248-012-9391-0. Epub 2012 Aug 24. AAPS J. 2012. PMID: 22918625 Free PMC article. Review.
-
A Novel Biotinylated Homotryptamine Derivative for Quantum Dot Imaging of Serotonin Transporter in Live Cells.Front Cell Neurosci. 2021 Nov 18;15:667044. doi: 10.3389/fncel.2021.667044. eCollection 2021. Front Cell Neurosci. 2021. PMID: 34867196 Free PMC article.
References
-
- Amara S.; Sonders M. S. (1998) Neurotransmitter transporters as molecular targets for addictive drugs. Drug Alcohol Depend. 51, 87–96. - PubMed
-
- Bowton E.; Saunders C.; Erreger K.; Sakrikar D.; Matthies H. J.; Sen N.; Jessen T.; Colbran R. J.; Caron M. G.; Javitch J. A.; Blakely R. D.; Galli A. (2010) Dysregulation of dopamine transporters via dopamine D2 autoreceptors triggers anomalous dopamine efflux associated with attention-deficit hyperactivity disorder. J. Neurosci. 30, 6048–6057. - PMC - PubMed
-
- Wong D. F.; Harris J. C.; Naidu S.; Yokoi F.; Marenco S.; Dannals R. F.; Ravert H. T.; Yaster M.; Evans A.; Rousset O.; Bryan R. N.; Gjedde A.; Kuhar M. J.; Breese G. R. (1996) Dopamine transporters are markedly reduced in Lesch-Nyhan disease in vivo. Proc. Natl. Acad. Sci. U.S.A. 93, 5539–5543. - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

