Enhancement of curcumin oral absorption and pharmacokinetics of curcuminoids and curcumin metabolites in mice
- PMID: 21968952
- PMCID: PMC3368221
- DOI: 10.1007/s00280-011-1749-y
Enhancement of curcumin oral absorption and pharmacokinetics of curcuminoids and curcumin metabolites in mice
Abstract
Purpose: Curcumin has shown a variety of biological activity for various human diseases including cancer in preclinical setting. Its poor oral bioavailability poses significant pharmacological barriers to its clinical application. Here, we established a practical nano-emulsion curcumin (NEC) containing up to 20% curcumin (w/w) and conducted the pharmacokinetics of curcuminoids and curcumin metabolites in mice.
Methods: This high loading NEC was formulated based on the high solubility of curcumin in polyethylene glycols (PEGs) and the synergistic enhancement of curcumin absorption by PEGs and Cremophor EL. The pharmacokinetics of curcuminoids and curcumin metabolites was characterized in mice using a LC-MS/MS method, and the pharmacokinetic parameters were determined using WinNonlin computer software.
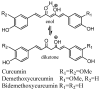
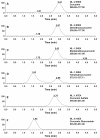
Results: A tenfold increase in the AUC (0→24h) and more than 40-fold increase in the C (max) in mice were observed after an oral dose of NEC compared with suspension curcumin in 1% methylcellulose. The plasma pharmacokinetics of its two natural congeners, demethoxycurcumin and bisdemethoxycurcumin, and three metabolites, tetrahydrocurcumin (THC), curcumin-O-glucuronide, and curcumin-O-sulfate, was characterized for the first time in mice after an oral dose of NEC.
Conclusion: This oral absorption enhanced NEC may provide a practical formulation to conduct the correlative study of the PK of curcuminoids and their pharmacodynamics, e.g., hypomethylation activity in vivo.
Figures




References
-
- Lopez-Lazaro M. Anticancer and carcinogenic properties of curcumin: considerations for its clinical development as a cancer chemopreventive and chemotherapeutic agent. Mol Nutr Food Res. 2008;52(Suppl 1):S103–S127. - PubMed
-
- Hsu CH, Cheng AL. Clinical studies with curcumin. Adv Exp Med Biol. 2007;595:471–480. - PubMed
-
- Pan MH, Huang TM, Lin JK. Biotransformation of curcumin through reduction and glucuronidation in mice. Drug Metab Dispos Biol Fate Chem. 1999;27:486–494. - PubMed
-
- Perkins S, Verschoyle RD, Hill K, Parveen I, Threadgill MD, Sharma RA, Williams ML, Steward WP, Gescher AJ. Chemopreventive efficacy and pharmacokinetics of curcumin in the min/+ mouse: a model of familial adenomatous polyposis. Cancer Epidemiol Biomarkers Prev. 2002;11:535–540. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

