Effect of Different Carriers on in vitro Permeation of Meloxicam through Rat Skin
- PMID: 21969742
- PMCID: PMC3178971
- DOI: 10.4103/0250-474X.84579
Effect of Different Carriers on in vitro Permeation of Meloxicam through Rat Skin
Abstract
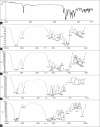
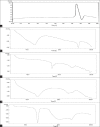
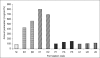
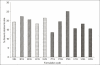
The ability of β-cyclodextrin, hydroxypropyl-β-cyclodextrin, polyvinyl pyrrolidone and urea to influence the percutaneous absorption of meloxicam through isolated rat skin was evaluated. Carrier complex were prepared by kneading method in 1:1 and 1:2 in molar ratios for β-cyclodextrin and hydroxypropyl-β-cyclodextrin and in 1:1, 1:3 and 1:5 in weight ratios for polyvinyl pyrrolidone and urea. The complexes were characterized by IR, DSC and evaluated for solubility, dissolution and skin permeability. The solubility, dissolution and permeability of meloxicam were enhanced by using the carriers. The influence of cyclodextrins, polyvinyl pyrrolidone and urea on in vitro permeation of meloxicam through rat skin was investigated by incorporation of prepared carrier complex in 1% carbopol gel. The prepared gel was evaluated for drug content, pH and viscosity and in vitro permeation. All the percutaneous parameters like flux (Jss), amount permeated (Q(6)), diffusivity (D), permeability coefficient (K(p)), partition coefficient (K) and release rate constant (k) were calculated statistically. In vitro permeation study showed the trend that the penetration flux and enhancement factor increases with increasing concentration of β-cyclodextrin and hydroxypropyl-β-cyclodextrin and then decrease dramatically in case of hydroxypropyl-β-cyclodextrin gel formulation with the increase to 1:2 ratio. Similar changes in pattern of permeation were also observed with polyvinyl pyrrolidone and urea carrier complex. These findings concluded that the carriers cyclodextrins, polyvinyl pyrrolidone and urea could be used as transdermal permeation enhancer in topical preparation of meloxicam.
Keywords: hydroxypropyl-β-cyclodextrin; in vitro release; meloxicam; permeation; polyvinyl pyrrolidone; urea; β-cyclodextrin.
Figures



Similar articles
-
Elastic liposomes bearing meloxicam-beta-cyclodextrin for transdermal delivery.Curr Drug Deliv. 2008 Jul;5(3):207-14. doi: 10.2174/156720108784911677. Curr Drug Deliv. 2008. PMID: 18673264
-
Meloxicam β-cyclodextrin transdermal gel: physicochemical characterization and in vitro dissolution and diffusion studies.Curr Drug Deliv. 2011 Jul;8(4):381-91. doi: 10.2174/156720111795767942. Curr Drug Deliv. 2011. PMID: 21453259
-
Influence of hydroxypropyl-beta-cyclodextrin on the transdermal permeation and skin accumulation of oxybenzone.Drug Dev Ind Pharm. 2002 Oct;28(9):1117-24. doi: 10.1081/ddc-120014578. Drug Dev Ind Pharm. 2002. PMID: 12455470
-
Mechanistic studies of the effect of hydroxypropyl-beta-cyclodextrin on in vitro transdermal permeation of corticosterone through hairless mouse skin.Int J Pharm. 2003 Mar 6;253(1-2):1-11. doi: 10.1016/s0378-5173(02)00625-7. Int J Pharm. 2003. PMID: 12593932
-
Evaluation of skin permeation and analgesic activity effects of carbopol lornoxicam topical gels containing penetration enhancer.ScientificWorldJournal. 2014;2014:127495. doi: 10.1155/2014/127495. Epub 2014 Jun 19. ScientificWorldJournal. 2014. PMID: 25045724 Free PMC article. Clinical Trial.
Cited by
-
Design, formulation, and physicochemical evaluation of periodontal propolis mucoadhesive gel.Dent Res J (Isfahan). 2016 Nov-Dec;13(6):484-493. doi: 10.4103/1735-3327.197037. Dent Res J (Isfahan). 2016. PMID: 28182054 Free PMC article.
-
Biodegradable microneedle patch for delivery of meloxicam for managing pain in cattle.PLoS One. 2022 Aug 2;17(8):e0272169. doi: 10.1371/journal.pone.0272169. eCollection 2022. PLoS One. 2022. PMID: 35917312 Free PMC article.
-
Design, Formulation, and Physicochemical Evaluation of Vaginal Cream Containing Eucalyptus camaldulensis, Viola odorata, and Mentha piperita extracts for Prevention and Treatment of Trichomoniasis.Int J Prev Med. 2019 Oct 9;10:179. doi: 10.4103/ijpvm.IJPVM_525_17. eCollection 2019. Int J Prev Med. 2019. PMID: 32133097 Free PMC article.
-
Design, formulation and evaluation of a mucoadhesive gel from Quercus brantii L. and coriandrum sativum L. as periodontal drug delivery.Adv Biomed Res. 2013 Mar 6;2:21. doi: 10.4103/2277-9175.108007. eCollection 2013. Adv Biomed Res. 2013. PMID: 23977649 Free PMC article.
-
Scl gene construction, expression and effect on hemangioma.Mol Biol Rep. 2013 Apr;40(4):3381-8. doi: 10.1007/s11033-012-2415-0. Epub 2012 Dec 28. Mol Biol Rep. 2013. PMID: 23271126
References
-
- Barry BW. Novel mechanisms and devices to enable successful transdermal drug delivery. Eur J Pharm Sci. 2001;14:101–14. - PubMed
-
- Williams AC, Barry BW. Penetration enhancers. Adv Drug Deliv Rev. 2004;56:603–18. - PubMed
-
- Nishihata T, Kamada A, Sakai K, Takahashi K, Matsumoto K, Shinozaki K, et al. Percutaneous absorption of diclofenac in rats and humans: Aqueous gel formulation. Int J Pharm. 1988;46:1–7.
-
- Loftsson T, Masson M. Cyclodextrins in topical drug formulations: Theory and practice. Int J Pharm. 2001;225:5–30. - PubMed
-
- Masson M, Loftsson T, Masson G, Stefansson E. Cyclodextrins as permeation enhancers: Some theoretical evaluations and in vitro testing. J Control Release. 1999;59:107–8. - PubMed
LinkOut - more resources
Full Text Sources
