Filopodia and adhesion in cancer cell motility
- PMID: 21975551
- PMCID: PMC3218609
- DOI: 10.4161/cam.5.5.17723
Filopodia and adhesion in cancer cell motility
Abstract
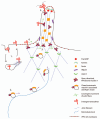
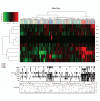
Slender bundled actin containing plasma membrane protrusions, called filopodia, are important for many essential cellular processes like cell adhesion, migration, angiogenesis and the formation of cell-cell contacts. In migrating cells, filopodia are the pioneers at the leading edge which probe the environment for cues. Integrins are cell surface adhesion receptors critically implicated in cell migration and they are transported actively to filopodia tips by an unconventional myosin, myosin-X. Integrin mediated adhesion stabilizes filopodia and promotes cell migration even though integrins are not essential for filopodia initiation. Myosin-X binds also PIP3 and this regulates its activation and localization to filopodia. Filopodia stimulate cell migration in many cell types and increased filopodia density has been described in cancer. Furthermore, several proteins implicated in filopodia formation, like fascin, are also relevant for cancer progression. To investigate this further, we performed a meta-analysis of the expression profiles of 10 filopodia-linked genes in human breast cancer. These data implicated that several different filopodia inducing genes may contribute in a collective manner to cancer progression and the high metastasis rates associated with basal-type breast carcinomas.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
