Achieving sufficient accrual to address the primary endpoint in phase III clinical trials from U.S. Cooperative Oncology Groups
- PMID: 21976533
- PMCID: PMC3977198
- DOI: 10.1158/1078-0432.CCR-11-1633
Achieving sufficient accrual to address the primary endpoint in phase III clinical trials from U.S. Cooperative Oncology Groups
Abstract
Purpose: Assessing impact of poor accrual on premature trial closure requires a relevant metric. We propose defining accrual sufficiency on apparent ability to address primary endpoints (PE) rather than attaining accrual targets.
Experimental design: All phase III trials open January 1, 1993, to December 31, 2002, by five U.S. oncology Clinical Trials Cooperative Groups (CTCG) were evaluated for accrual sufficiency and scientific results. Sufficient accrual included meeting accrual target, CTCGs documentation attesting adequate accrual, or conclusive results at interim analysis; insufficient accrual included poor accrual as cited closure reason or other reasons rendering a trial unable to address its primary endpoints. Closure rates based on our accrual sufficiency definition are compared with rates of meeting accrual targets and addressing the primary endpoints. A percentage of target accrual above which trials commonly answer the intended scientific question was identified to serve as an alternative to meeting full target accrual in designating accrual success.
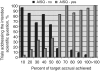
Results: Of 238 eligible trials, 158 (66%) closed with sufficient accrual. Among 80 trials with insufficient accrual, 70 (29%) closed specifically because of poor accrual. Inadequate accrual rates are overemphasized when defining accrual success solely by meeting accrual targets. Nearly 75% of trials conclusively addressed the primary endpoints with positive results in 39% of trials. Exceeding 80% of target accrual serves as a reliable proxy for answering the intended scientific question.
Conclusions: Approximately one third of phase III trials closed with insufficient accrual to address the primary endpoints, primarily due to poor accrual. Defining accrual sufficiency broader than meeting accrual targets represents a fairer account of trial closures.
© 2011 AACR.
Conflict of interest statement
Figures


Comment in
-
The importance of doing trials right while doing the right trials.Clin Cancer Res. 2012 Jan 1;18(1):3-5. doi: 10.1158/1078-0432.CCR-11-2586. Epub 2011 Nov 9. Clin Cancer Res. 2012. PMID: 22072734 Free PMC article.
References
-
- Institute of Medicine. A national cancer clinical trials system for the 21st century: reinvigorating the NCI cooperative group program. Washington, DC: IOM (Institute of Medicine) The National Academies Press; 2010. - PubMed
-
- Collins JF, Williford WO, Weiss DG, Bingham SF, Klett CJ. Planning patient recruitment: fantasy and reality. Stat Med. 1984;3:435–43. - PubMed
Publication types
MeSH terms
Grants and funding
- U24-CA-114732/CA/NCI NIH HHS/United States
- P30 CA044579/CA/NCI NIH HHS/United States
- U10CA-25224/CA/NCI NIH HHS/United States
- U10 CA069651/CA/NCI NIH HHS/United States
- U10CA-37377/CA/NCI NIH HHS/United States
- U10 CA069974/CA/NCI NIH HHS/United States
- R01 CA118232/CA/NCI NIH HHS/United States
- U10 CA025224/CA/NCI NIH HHS/United States
- U10CA-12027/CA/NCI NIH HHS/United States
- U10 CA180868/CA/NCI NIH HHS/United States
- U10CA-69974/CA/NCI NIH HHS/United States
- U10 CA012027/CA/NCI NIH HHS/United States
- U24 CA114732/CA/NCI NIH HHS/United States
- U10 CA037377/CA/NCI NIH HHS/United States
- U10CA-69651/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources

