Enteral omega-3 fatty acid, gamma-linolenic acid, and antioxidant supplementation in acute lung injury
- PMID: 21976613
- PMCID: PMC3770348
- DOI: 10.1001/jama.2011.1435
Enteral omega-3 fatty acid, gamma-linolenic acid, and antioxidant supplementation in acute lung injury
Erratum in
- JAMA. 2012 Feb 8;307(6):563
Abstract
Context: The omega-3 (n-3) fatty acids docosahexaenoic acid and eicosapentaenoic acid, along with γ-linolenic acid and antioxidants, may modulate systemic inflammatory response and improve oxygenation and outcomes in patients with acute lung injury.
Objective: To determine if dietary supplementation of these substances to patients with acute lung injury would increase ventilator-free days to study day 28.
Design, setting, and participants: The OMEGA study, a randomized, double-blind, placebo-controlled, multicenter trial conducted from January 2, 2008, through February 21, 2009. Participants were 272 adults within 48 hours of developing acute lung injury requiring mechanical ventilation whose physicians intended to start enteral nutrition at 44 hospitals in the National Heart, Lung, and Blood Institute ARDS Clinical Trials Network. All participants had complete follow-up.
Interventions: Twice-daily enteral supplementation of n-3 fatty acids, γ-linolenic acid, and antioxidants compared with an isocaloric control. Enteral nutrition, directed by a protocol, was delivered separately from the study supplement.
Main outcome measure: Ventilator-free days to study day 28.
Results: The study was stopped early for futility after 143 and 129 patients were enrolled in the n-3 and control groups. Despite an 8-fold increase in plasma eicosapentaenoic acid levels, patients receiving the n-3 supplement had fewer ventilator-free days (14.0 vs 17.2; P = .02) (difference, -3.2 [95% CI, -5.8 to -0.7]) and intensive care unit-free days (14.0 vs 16.7; P = .04). Patients in the n-3 group also had fewer nonpulmonary organ failure-free days (12.3 vs 15.5; P = .02). Sixty-day hospital mortality was 26.6% in the n-3 group vs 16.3% in the control group (P = .054), and adjusted 60-day mortality was 25.1% and 17.6% in the n-3 and control groups, respectively (P = .11). Use of the n-3 supplement resulted in more days with diarrhea (29% vs 21%; P = .001).
Conclusions: Twice-daily enteral supplementation of n-3 fatty acids, γ-linolenic acid, and antioxidants did not improve the primary end point of ventilator-free days or other clinical outcomes in patients with acute lung injury and may be harmful.
Trial registration: clinicaltrials.gov Identifier: NCT00609180.
Figures


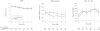
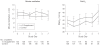

Comment in
-
Pharmaconutrition in acute lung injury.JAMA. 2011 Oct 12;306(14):1599-600. doi: 10.1001/jama.2011.1470. Epub 2011 Oct 5. JAMA. 2011. PMID: 21976612 No abstract available.
-
Supplementation in acute lung injury.JAMA. 2012 Jan 11;307(2):144; author reply 145-6. doi: 10.1001/jama.2011.1982. JAMA. 2012. PMID: 22235078 No abstract available.
-
Supplementation in acute lung injury.JAMA. 2012 Jan 11;307(2):144-5; author reply 145-6. doi: 10.1001/jama.2011.1983. JAMA. 2012. PMID: 22235079 No abstract available.
-
Supplementation in acute lung injury.JAMA. 2012 Jan 11;307(2):145; author reply 145-6. doi: 10.1001/jama.2011.1984. JAMA. 2012. PMID: 22235080 No abstract available.
-
ω-3 fatty acids, γ-linolenic acid, and antioxidants: immunomodulators or inert dietary supplements?Crit Care. 2012 Nov 23;16(6):325. doi: 10.1186/cc11863. Crit Care. 2012. PMID: 23176247 Free PMC article.
References
-
- Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med. 2000;342(18):1334–1349. - PubMed
-
- Abraham E. Coagulation abnormalities in acute lung injury and sepsis. Am J Respir Cell Mol Biol. 2000;22(4):401–404. - PubMed
-
- Gust R, Kozlowski JK, Stephenson AH, Schuster DP. Role of cyclooxygenase-2 in oleic acid-induced acute lung injury. Am J Respir Crit Care Med. 1999;160(4):1165–1170. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- N01 HR056168/HL/NHLBI NIH HHS/United States
- N01 HR056174/HL/NHLBI NIH HHS/United States
- N01 HR056169/HL/NHLBI NIH HHS/United States
- N01 HR056179/HL/NHLBI NIH HHS/United States
- N01 HR056165/HL/NHLBI NIH HHS/United States
- N01 HR056175/HL/NHLBI NIH HHS/United States
- N01 HR056176/HL/NHLBI NIH HHS/United States
- HHSN268200536176C/PHS HHS/United States
- N01 HR056166/HL/NHLBI NIH HHS/United States
- HHSN268200536179C/PHS HHS/United States
- N01 HR056173/HL/NHLBI NIH HHS/United States
- N01 HR056170/HR/NHLBI NIH HHS/United States
- N01 HR056167/HL/NHLBI NIH HHS/United States
- HHSN268200536165C/PHS HHS/United States
- N01 HR056172/HL/NHLBI NIH HHS/United States
- N01 HR056171/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

