Biodegradable ocular inserts for sustained delivery of brimonidine tartarate: preparation and in vitro/in vivo evaluation
- PMID: 21979886
- PMCID: PMC3225539
- DOI: 10.1208/s12249-011-9701-3
Biodegradable ocular inserts for sustained delivery of brimonidine tartarate: preparation and in vitro/in vivo evaluation
Abstract
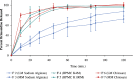
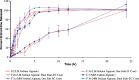
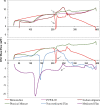
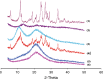
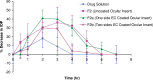
The bioavailability of therapeutic agents from eye drops is usually limited due to corneal barrier functions and effective eye protective mechanisms. Therefore, the current study aims to enhance ocular bioavailability of brimonidine, a potent antiglaucoma drug, through the preparation of ocular inserts. Solvent casting technique was employed to prepare the inserts using polyvinylpyrrolidone K-90 (PVP K-90) as film-forming polymer blended with different viscosity grades of bioadhesive polymers namely hydroxypropyl methycellulose, carbopol, sodium alginate, and chitosan. The prepared ocular inserts were evaluated for various physicochemical parameters, swelling behavior, and in vitro release patterns. Sodium alginate-based ocular inserts revealed the most sustainment in drug release (99% at 6 h), so it was selected for further modifications via coating it, on one side or dual sides, using hydrophobic film composed of either ethylcellulose or Eudragit RSPO. The obtained in vitro release results for the modified ocular inserts revealed that ethylcellulose is superior to Eudragit RSPO in terms of brimonidine release sustainment effect. Ocular inserts composed of 7% PVP K-90, 1.5% low molecular weight sodium alginate with or without ethylcellulose coat were able to sustain the in vitro release of brimonidine. Their therapeutic efficacy regarding intraocular pressure (IOP) lowering effect when inserted in albino rabbits eyes showed superior sustainment effect compared with that of brimonidine solution. Furthermore, due to both the mucoadhesive property and the drug sustainment effect, the one-side-coated ocular insert showed more IOP lowering effect compared with that of its non-coated or dual-side-coated counterpart.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

