Characterization of tissue biomechanics and mechanical signaling in uterine leiomyoma
- PMID: 21983114
- PMCID: PMC3576015
- DOI: 10.1016/j.matbio.2011.09.001
Characterization of tissue biomechanics and mechanical signaling in uterine leiomyoma
Abstract
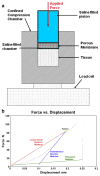
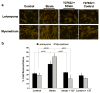
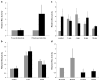
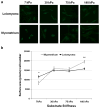
Leiomyoma are common tumors arising within the uterus that feature excessive deposition of a stiff, disordered extracellular matrix (ECM). Mechanical stress is a critical determinant of excessive ECM deposition and increased mechanical stress has been shown to be involved in tumorigenesis. Here we tested the viscoelastic properties of leiomyoma and characterized dynamic and static mechanical signaling in leiomyoma cells using three approaches, including measurement of active RhoA. We found that the peak strain and pseudo-dynamic modulus of leiomyoma tissue was significantly increased relative to matched myometrium. In addition, leiomyoma cells demonstrated an attenuated response to applied cyclic uniaxial strain and to variation in substrate stiffness, relative to myometrial cells. However, on a flexible pronectin-coated silicone substrate, basal levels and lysophosphatidic acid-stimulated levels of activated RhoA were similar between leiomyoma and myometrial cells. In contrast, leiomyoma cells plated on a rigid polystyrene substrate had elevated levels of active RhoA, compared to myometrial cells. The results indicate that viscoelastic properties of the ECM of leiomyoma contribute significantly to the tumor's inherent stiffness and that leiomyoma cells have an attenuated sensitivity to mechanical cues. The findings suggest there may be a fundamental alteration in the communication between the external mechanical environment (extracellular forces) and reorganization of the actin cytoskeleton mediated by RhoA in leiomyoma cells. Additional research will be needed to elucidate the mechanism(s) responsible for the attenuated mechanical signaling in leiomyoma cells.
Published by Elsevier B.V.
Conflict of interest statement
None.
Figures

Similar articles
-
Simvastatin ameliorates altered mechanotransduction in uterine leiomyoma cells.Am J Obstet Gynecol. 2020 Nov;223(5):733.e1-733.e14. doi: 10.1016/j.ajog.2020.05.012. Epub 2020 May 15. Am J Obstet Gynecol. 2020. PMID: 32417359 Free PMC article.
-
Mechanical homeostasis is altered in uterine leiomyoma.Am J Obstet Gynecol. 2008 Apr;198(4):474.e1-11. doi: 10.1016/j.ajog.2007.11.057. Am J Obstet Gynecol. 2008. PMID: 18395046 Free PMC article.
-
Mechanical stiffness augments ligand-dependent progesterone receptor B activation via MEK 1/2 and Rho/ROCK-dependent signaling pathways in uterine fibroid cells.Fertil Steril. 2021 Jul;116(1):255-265. doi: 10.1016/j.fertnstert.2020.12.011. Epub 2021 Mar 3. Fertil Steril. 2021. PMID: 33676751
-
Mechanical Signaling and Extracellular Matrix in Uterine Fibroids.Semin Reprod Med. 2017 Nov;35(6):487-493. doi: 10.1055/s-0037-1607268. Epub 2017 Nov 3. Semin Reprod Med. 2017. PMID: 29100236 Review. No abstract available.
-
Extracellular matrix in uterine leiomyoma pathogenesis: a potential target for future therapeutics.Hum Reprod Update. 2018 Jan 1;24(1):59-85. doi: 10.1093/humupd/dmx032. Hum Reprod Update. 2018. PMID: 29186429 Review.
Cited by
-
Is ulipristal acetate the new drug of choice for the medical management of uterine fibroids? Res ipsa loquitur?Womens Health (Lond). 2017 Dec;13(3):98-105. doi: 10.1177/1745505717740218. Epub 2017 Nov 6. Womens Health (Lond). 2017. PMID: 29105584 Free PMC article. Review.
-
Literature Review on the Role of Uterine Fibroids in Endometrial Function.Reprod Sci. 2018 May;25(5):635-643. doi: 10.1177/1933719117725827. Epub 2017 Aug 22. Reprod Sci. 2018. PMID: 28826369 Free PMC article. Review.
-
Bromodomain-Containing Protein 9 Regulates Signaling Pathways and Reprograms the Epigenome in Immortalized Human Uterine Fibroid Cells.Int J Mol Sci. 2024 Jan 11;25(2):905. doi: 10.3390/ijms25020905. Int J Mol Sci. 2024. PMID: 38255982 Free PMC article.
-
Uterine fibroids: pathogenesis and interactions with endometrium and endomyometrial junction.Obstet Gynecol Int. 2013;2013:173184. doi: 10.1155/2013/173184. Epub 2013 Sep 12. Obstet Gynecol Int. 2013. PMID: 24163697 Free PMC article. Review.
-
NAV3, a Tumor Suppressor Gene, Is Decreased in Uterine Leiomyoma Tissue and Cells.Reprod Sci. 2020 Mar;27(3):925-934. doi: 10.1007/s43032-019-00096-3. Epub 2020 Jan 1. Reprod Sci. 2020. PMID: 32046415 Free PMC article. Clinical Trial.
References
-
- Aleem FA, Predanic M. The hemodynamic effect of GnRH agonist therapy on uterine leiomyoma vascularity: a prospective study using transvaginal color Doppler sonography. Gynecol Endocrinol. 1995;9:253–258. - PubMed
-
- Alenghat FJ, Ingber DE. Mechanotransduction: all signals point to cytoskeleton, matrix, and integrins. Sci STKE. 2002;(119):pe6. - PubMed
-
- Alexopoulos LG, Setton LA, Guilak F. The biomechanical role of the chondrocyte pericellular matrix in articular cartilage. Acta Biomater. 2005;1:317–325. - PubMed
-
- Ateshian GA, Soltz MA, Mauck RL, Basalo IM, Hung CT, Lai WM. The role of osmotic pressure and tension-compression nonlinearity in the frictional response of articular cartilage. Transp Porous Media. 2003;50:5–33.
-
- Behera MA, Feng L, Yonish B, Catherino W, Jung SH, Leppert P. Thrombospondin-1 and thrombospondin-2 mRNA and TSP-1 and TSP-2 protein expression in uterine fibroids and correlation to the genes COL1A1 and COL3A1 and to the collagen cross-link hydroxyproline. Reprod Sci. 2007;14:63–76. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

